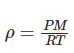Ideal gas law in terms of density
Click For Summary
Homework Help Overview
The discussion revolves around the ideal gas law and its application in terms of density, specifically focusing on the relationship between pressure, density, and height in a fluid context.
Discussion Character
- Exploratory, Mathematical reasoning, Assumption checking
Approaches and Questions Raised
- Participants explore the ideal gas law equation and its rearrangement to relate pressure and density. There are attempts to integrate a derived equation involving pressure and height, with some participants expressing uncertainty about integration techniques.
Discussion Status
Participants are actively engaging with the problem, discussing integration methods and the implications of constants in the equations. Some guidance has been offered regarding integration, but there is still uncertainty and a lack of consensus on the next steps.
Contextual Notes
There are indications of varying levels of mathematical knowledge among participants, particularly regarding integration and differentiation, which may affect their ability to progress in solving the problem.
Similar threads
- · Replies 9 ·
- · Replies 2 ·
- · Replies 3 ·
- · Replies 2 ·
- · Replies 3 ·
- · Replies 3 ·
- · Replies 2 ·
- · Replies 12 ·