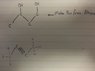
Discussion Overview
The discussion revolves around the synthesis of an alcohol from an alkyne, exploring various methods and reactions in organic chemistry. Participants share their approaches and reasoning related to hydration reactions, halide substitutions, and the conversion of alkynes to alkenes.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
- One participant suggests using a hydration reaction to convert an alkyne to an alcohol by mixing it with water.
- Another participant proposes hydrating the alkyne and then substituting a hydroxyl group with a halide using PBr3, but later revises their suggestion, indicating a misunderstanding about the resulting structure.
- A third participant outlines a multi-step process involving the conversion of the alkyne to an alkene using Na/liq.NH3, followed by the use of NBS to obtain allyl bromide, which is then reacted with aqueous NaOH to yield the desired alcohol.
- Another participant believes that mixing an alkene with water could yield the desired alcohol, specifically mentioning the formation of 1,3-butanediol.
Areas of Agreement / Disagreement
Participants present multiple competing views on how to synthesize the alcohol, with no consensus reached on the best method. Some approaches are challenged or revised, indicating uncertainty in the proposed reactions.
Contextual Notes
Some participants express uncertainty about the accuracy of their chemical structures and reaction mechanisms, and there are indications of missing assumptions regarding the steps involved in the synthesis.
Who May Find This Useful
This discussion may be useful for students or practitioners in organic chemistry looking for various methods of alcohol synthesis from alkynes, as well as those interested in the nuances of reaction mechanisms and potential pitfalls in organic reactions.