utsav55
- 14
- 0
What will be the % of Enol form of this compound?
99% or 1%? Also give reason.
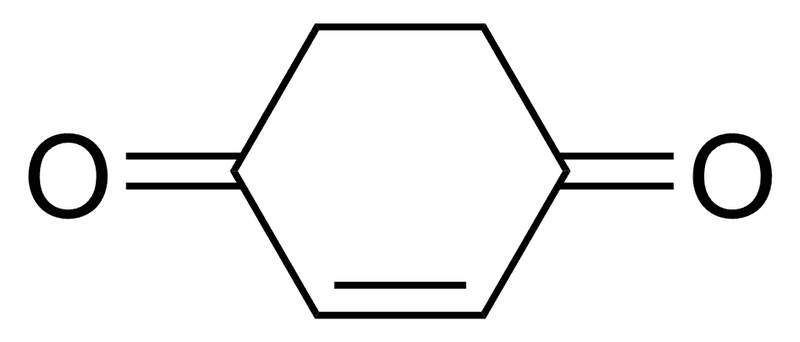
Thanks
99% or 1%? Also give reason.
Thanks
The discussion revolves around the percentage of the enol form of a specific compound, with participants debating the stability of the keto versus enol forms. The scope includes theoretical considerations of tautomerism and aromaticity.
Participants express differing opinions on the stability and percentage of the enol form, indicating that multiple competing views remain without a consensus.
There are unresolved assumptions regarding the definitions of aromaticity and the specific compound in question, as well as the implications of Gibbs free energy on the equilibrium percentages.