- #1
johnconnor
- 62
- 0
I would appreciate if we could discuss the questions together. It's a very interesting question indeed.
Question:
The singly ionised hydrogen molecule [tex]H_2^+[/tex] can be regarded as an electron of charge -e moving in the field due to two protons, each of charge +e. The protons, which can be assumed to be stationary, are a distance r apart, as shown in Fig. SM11.1. Figure SM11.2 shows how the total electrostatic energy E of the ionised molecule in its state of lowest energy depends on the separation r of the protons. The zero of energy is the situation in which the electron and the protons are all infinitely far away from each other.
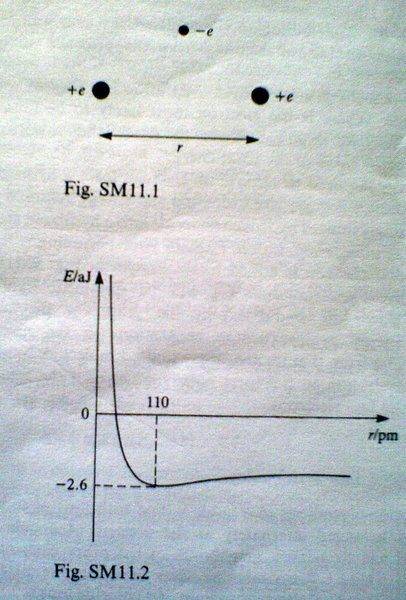
(a) The graph in Fig SM11.2 can be considered as the summation of a positive energy term and a negative energy term. Identify the origins of these terms.
(b) What is the physical reason for the fact that, for small values of r, the graph has a large negative gradient?
(c) What is the physical interpretation of the fact that the curve shows a minimum energy at a particular value of r (110pm)?
(d) What is the physical interpretation of the fact that the minimum value of the total energy is -2.6aJ?
(e) calculate the contribution to the total energy at a proton-proton separation of 110pm resulting from the presence of the electron.
(f) Suppose that the proton-proton separation is increased to a very large value. Where is the electron most likely to be found? Hence, explain why for large values of r, the curve tends towards a negarive (non-zero) value of the total energy.
(g) The energy required to remove an electron from a hydrogen atom (the ionisation energy) is 2.2aJ. Find the energy required to dissociate the singly ionised hydrogen molecule into a hydrogen atom and a proton at infinite separation. Explain your reasoning.
(h) If a second election is added to the system of Fig. SM11.1, the neutral hydrogen molecule H2 will be obtained. Explain qualitatively how you would expect the graph of the total electrostatic energy against proton separation for the neutral molecule to differ from that for the singly ionised molecule. Neglect the small contribution to the total energy caused by the mutual repulsion of the two electrons.
Attempt
Please do correct and/or refine the answers I have given here. Forgive me if my mistakes are elementary:
(a) Protons and electrons.
(b) Repulsion force, which in this case is taken to be positive from -dU/dr (or in this case dE)
(c) Presence of electron produces a minimum net force acting on the protons and electron.
(d) see (c)
(e) proton-electron coulombic potential: [tex]\frac{+Q.-e}{4\pi \varepsilon _0 (110pm)}= -2.097aJ[/tex] Therefore -2.6aJ + -2.097aJ = -4.7aJ.
(f) Near to one of the protons. Otherwise there wouldn't be a negative energy value as r tends to a large value. So, the ion will kinda look like a hydrogen proton with a distant proton neighbour. (How do I properly phrase this? Is there any better way to explain this?)
(g) 1st IE for H = 2.2aJ (as previously mentioned)
-2.6aJ = energy to separate all particles to infinity.
Hence, energy required to dissociate the singly ionised hydrogen molecule into a hydrogen atom and a proton at infinite separation = 2.6 - 2.2 = 0.4aJ
(h) Beats me.
Please help me out on this. I'll spam a bit on this forum to reach 10 posts and I'll post the photo over here. Thank you! Your help is greatly appreciated!
Question:
The singly ionised hydrogen molecule [tex]H_2^+[/tex] can be regarded as an electron of charge -e moving in the field due to two protons, each of charge +e. The protons, which can be assumed to be stationary, are a distance r apart, as shown in Fig. SM11.1. Figure SM11.2 shows how the total electrostatic energy E of the ionised molecule in its state of lowest energy depends on the separation r of the protons. The zero of energy is the situation in which the electron and the protons are all infinitely far away from each other.
(a) The graph in Fig SM11.2 can be considered as the summation of a positive energy term and a negative energy term. Identify the origins of these terms.
(b) What is the physical reason for the fact that, for small values of r, the graph has a large negative gradient?
(c) What is the physical interpretation of the fact that the curve shows a minimum energy at a particular value of r (110pm)?
(d) What is the physical interpretation of the fact that the minimum value of the total energy is -2.6aJ?
(e) calculate the contribution to the total energy at a proton-proton separation of 110pm resulting from the presence of the electron.
(f) Suppose that the proton-proton separation is increased to a very large value. Where is the electron most likely to be found? Hence, explain why for large values of r, the curve tends towards a negarive (non-zero) value of the total energy.
(g) The energy required to remove an electron from a hydrogen atom (the ionisation energy) is 2.2aJ. Find the energy required to dissociate the singly ionised hydrogen molecule into a hydrogen atom and a proton at infinite separation. Explain your reasoning.
(h) If a second election is added to the system of Fig. SM11.1, the neutral hydrogen molecule H2 will be obtained. Explain qualitatively how you would expect the graph of the total electrostatic energy against proton separation for the neutral molecule to differ from that for the singly ionised molecule. Neglect the small contribution to the total energy caused by the mutual repulsion of the two electrons.
Attempt
Please do correct and/or refine the answers I have given here. Forgive me if my mistakes are elementary:
(a) Protons and electrons.
(b) Repulsion force, which in this case is taken to be positive from -dU/dr (or in this case dE)
(c) Presence of electron produces a minimum net force acting on the protons and electron.
(d) see (c)
(e) proton-electron coulombic potential: [tex]\frac{+Q.-e}{4\pi \varepsilon _0 (110pm)}= -2.097aJ[/tex] Therefore -2.6aJ + -2.097aJ = -4.7aJ.
(f) Near to one of the protons. Otherwise there wouldn't be a negative energy value as r tends to a large value. So, the ion will kinda look like a hydrogen proton with a distant proton neighbour. (How do I properly phrase this? Is there any better way to explain this?)
(g) 1st IE for H = 2.2aJ (as previously mentioned)
-2.6aJ = energy to separate all particles to infinity.
Hence, energy required to dissociate the singly ionised hydrogen molecule into a hydrogen atom and a proton at infinite separation = 2.6 - 2.2 = 0.4aJ
(h) Beats me.
Please help me out on this. I'll spam a bit on this forum to reach 10 posts and I'll post the photo over here. Thank you! Your help is greatly appreciated!
Last edited:
