Discussion Overview
The discussion revolves around calculating the energy required to move a volume within a rotating gas, specifically focusing on the dynamics of pressure, temperature, and torque in relation to different shapes of the moving volume and gas densities. The participants explore theoretical implications and mathematical relationships without reaching a consensus.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
- Mathematical reasoning
Main Points Raised
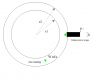
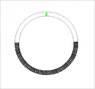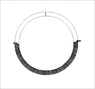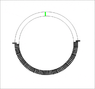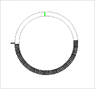
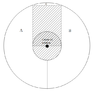
- One participant proposes calculating the energy needed for a black volume moving from r2 to r1 in a rotating gas, suggesting that the temperature of the gas remains constant during rotation.
- Another participant suggests that if the height (h) of the black volume is small compared to r1 and r2, the effects of rotation can be neglected, leading to similar average pressures inside and outside the volume.
- It is noted that the shape of the black volume affects the energy calculation, with a circular sector shape allowing for a fixed fraction of volume removal that does not depend on rotation.
- Concerns are raised about how different shapes of the black volume might alter the required energy, with one participant questioning the mechanical implications of a non-rotating volume in a rotating gas.
- Participants discuss the relationship between torque and gas density, with one questioning how different densities of gas would affect torque on a wall in a rotating system.
- There is a clarification that temperature relates to the unordered kinetic energy of gas, and that the total velocity of the gas is irrelevant to the energy calculations.
- One participant expresses confusion about the mechanical feasibility of moving a block into a turning wheel without affecting the wheel's integrity.
- Another participant suggests that numerical simulations may be necessary to fully understand the complex interactions of pressure, temperature, and density in the system.
Areas of Agreement / Disagreement
Participants express various viewpoints and uncertainties regarding the effects of rotation, shape, and density on energy calculations and torque. No consensus is reached on the implications of these factors or the mechanical feasibility of the proposed scenarios.
Contextual Notes
Limitations include unresolved assumptions about the effects of rotation on temperature and pressure, the dependence of calculations on the shape of the black volume, and the complexity of interactions that may require numerical simulations for clarification.
Who May Find This Useful
This discussion may be of interest to those studying fluid dynamics, thermodynamics, or mechanical engineering, particularly in contexts involving rotating systems and energy calculations in gases.