Discussion Overview
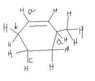
The discussion revolves around the stability of allylic carbanions formed from the cleavage of C-H bonds, as illustrated in a provided diagram. Participants explore the implications of resonance stabilization in different positions of the carbanions.
Discussion Character
- Homework-related, Conceptual clarification, Debate/contested
Main Points Raised
- Some participants suggest that cleavage of a C-H bond leads to the formation of a carbanion, with the stability influenced by the electronegativity of carbon compared to hydrogen.
- There is mention of two allylic carbanions (a and b) being formed, with a focus on determining which is more stable due to resonance stabilization.
- One participant notes that cleavage of position 'a' results in a vinylic carbanion, while position 'b' is associated with a more stable allylic carbanion.
- A later reply corrects an earlier statement, indicating that positions 'b' and 'd' produce allylic carbanions, prompting a question about their relative stability.
Areas of Agreement / Disagreement
Participants express differing views on which allylic carbanion is the most stable, indicating that the discussion remains unresolved with multiple competing perspectives.
Contextual Notes
Participants have not fully clarified the assumptions regarding the stability of the carbanions or the specific definitions of resonance stabilization being applied.