Discussion Overview
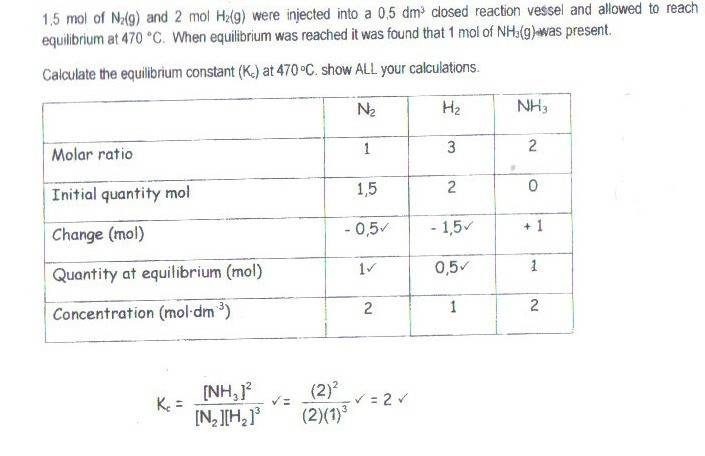
The discussion revolves around understanding the equilibrium constant table method, specifically focusing on how to interpret changes in moles of reactants and products in a chemical reaction. Participants explore the relationships between initial quantities, changes, and equilibrium quantities in the context of a reaction involving ammonia (NH3), nitrogen (N2), and hydrogen (H2).
Discussion Character
- Technical explanation
- Conceptual clarification
- Homework-related
Main Points Raised
- One participant expresses confusion about the "Change (mol)" in the equilibrium table and seeks clarification.
- Another participant suggests using molar ratios to determine how many moles of N2 and H2 are needed to produce 1 mole of NH3.
- A participant explains that to produce 2 molecules of NH3, 1 molecule of N2 is required, leading to the conclusion that 1/2 mole of N2 is needed for 1 mole of NH3.
- There is a discussion about how to calculate the change in moles, with one participant confirming that the mole ratios for N2 and H2 relative to NH3 are 0.5 and 1.5, respectively, and noting that the signs are negative for reactants being consumed.
- A question is raised about the implications of a negative change for NH3, suggesting that it would indicate a reverse reaction, with reactants increasing.
- Another participant agrees that initial NH3 would be necessary for the reverse reaction to occur.
Areas of Agreement / Disagreement
Participants generally agree on the relationships between moles of reactants and products, but there is some uncertainty regarding the implications of negative changes and the conditions required for reverse reactions.
Contextual Notes
The discussion does not resolve the broader implications of changes in equilibrium or the specific conditions under which reactions may reverse, leaving these aspects open for further exploration.