orome23
- 3
- 0
I have been working on an idea for a new energy source that I would like to get some input on. I took physics in college about 10 years ago, so I have a general understanding of the concepts, but I am definitely not an expert on thermodynamics.
Here is my idea:
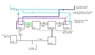
1. Use a heat pump(ground source, or air source in the right climate), to concentrate heat.
2. Use the concentrated heat in a boiler to generate steam.
3. Use the steam to drive a piston steam engine.
4. The steam engine then drives an electric generator.
5. Hopefully, the generator would produce more electricity than the heat pump and associated equipment need.
6. The exhausted steam from the engine is collected in a condenser, cooled to just below it's boiling point and injected back into the boiler. Well water would be used to provide cooling in the condenser, and then be routed to the heat pump to recycle the heat. This would increase the temperature of the water entering the heat pump and hopefully improve it's coefficient of performance(COP).
At first glance this looks like a perpetual motion machine. I would argue that it isn't, because the system will take heat from an external source(the ground, air, or anything else that is warm enough), use a heat pump to concentrate it, and turn it into electricity. For example, in a ground source version the water going back into the well, will be colder than it came out.
Another idea I have to improve efficiency is to use something other than water to produce the vapor(steam) that powers the engine.
Unfortunately my knowledge of heat pumps and steam engines is limited so I have a few questions:
1. How high of a COP can a ground source heat pump achieve? It would only be used for generating heat and not for cooling so I would think that it could be more efficient than heat pumps that are used in homes or businesses.
2. What sort of efficiency can really be obtained from a multi expansion chamber, piston steam engine?
3. What happens to the performance of a steam engine when a liquid other than water is used? Does using a liquid with a lower heat of vaporization affect the performance of the engine.
4. How high of a temperature can you achieve with a heat pump?
5. Is there something better than a steam engine to convert an external heat source into electricity?
Using the limited information I could find online I came up with these estimates for efficiency:
Input Heat Pump COP Boiler Efficiency Steam Engine Generator Inverter Output
1 unit 5.5 .95 .34 .9 .9 1.439
I think this idea is either genius or ridiculous so I would love to hear any opinions or suggestions that people may have, even if you think it's ridiculous.
Thanks,
Ned
P.S. I am working on a drawing to better explain, I will post it when it is finished.
Here is my idea:
1. Use a heat pump(ground source, or air source in the right climate), to concentrate heat.
2. Use the concentrated heat in a boiler to generate steam.
3. Use the steam to drive a piston steam engine.
4. The steam engine then drives an electric generator.
5. Hopefully, the generator would produce more electricity than the heat pump and associated equipment need.
6. The exhausted steam from the engine is collected in a condenser, cooled to just below it's boiling point and injected back into the boiler. Well water would be used to provide cooling in the condenser, and then be routed to the heat pump to recycle the heat. This would increase the temperature of the water entering the heat pump and hopefully improve it's coefficient of performance(COP).
At first glance this looks like a perpetual motion machine. I would argue that it isn't, because the system will take heat from an external source(the ground, air, or anything else that is warm enough), use a heat pump to concentrate it, and turn it into electricity. For example, in a ground source version the water going back into the well, will be colder than it came out.
Another idea I have to improve efficiency is to use something other than water to produce the vapor(steam) that powers the engine.
Unfortunately my knowledge of heat pumps and steam engines is limited so I have a few questions:
1. How high of a COP can a ground source heat pump achieve? It would only be used for generating heat and not for cooling so I would think that it could be more efficient than heat pumps that are used in homes or businesses.
2. What sort of efficiency can really be obtained from a multi expansion chamber, piston steam engine?
3. What happens to the performance of a steam engine when a liquid other than water is used? Does using a liquid with a lower heat of vaporization affect the performance of the engine.
4. How high of a temperature can you achieve with a heat pump?
5. Is there something better than a steam engine to convert an external heat source into electricity?
Using the limited information I could find online I came up with these estimates for efficiency:
Input Heat Pump COP Boiler Efficiency Steam Engine Generator Inverter Output
1 unit 5.5 .95 .34 .9 .9 1.439
I think this idea is either genius or ridiculous so I would love to hear any opinions or suggestions that people may have, even if you think it's ridiculous.
Thanks,
Ned
P.S. I am working on a drawing to better explain, I will post it when it is finished.