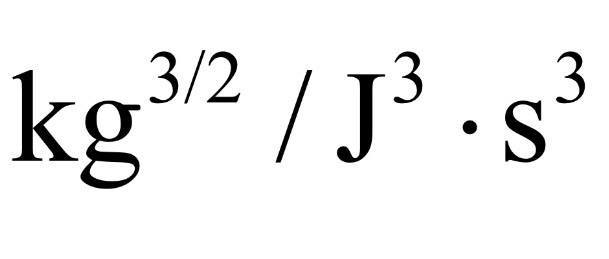Discussion Overview
The discussion revolves around the conversion of density of states to electron volts (eV), specifically addressing the dimensional analysis and unit conversion involved in this process.
Discussion Character
- Technical explanation
- Mathematical reasoning
Main Points Raised
- One participant suggests that multiplying by e^(3/2) can facilitate the conversion to eV, but seeks clarification on the reasoning behind this.
- Another participant points out that converting units with dimensions of [J^-3] to [eV] is not possible, questioning if the intent is to convert to [eV^-3] instead.
- A subsequent response confirms the intention to convert to [eV^-3].
- Further, a participant introduces a comparison involving the conversion of units from cubic miles to cubic meters, drawing a parallel to converting from cubic joules to cubic electron volts.
- Lastly, a participant expresses gratitude for the assistance provided in the discussion.
Areas of Agreement / Disagreement
The discussion contains multiple viewpoints regarding the conversion process and the appropriate units, indicating that there is no consensus on the method to achieve the desired conversion.
Contextual Notes
Participants have not fully resolved the assumptions regarding the conversion factors and the dimensionality of the units involved, leaving some steps in the conversion process unclear.