yerpo
- 24
- 1
I appologize in advance if this problem will be awkwardly presented, but I'm a biologist, with no talent for physics or mathematics whatsoever. Here goes...
At my institution, we are researching communication of certain bugs, which use low-frequency, substrate-borne vibrations as signals. Those signals are so well tuned to the physical properties of plants the bugs live on, that they transmit with very little attenuation, and so they rebound (probably) several times from stem boundaries before dying down. As far as we know, those are surface vibrations, transmitted in the form of bending waves. I measured the amplitude of artificially-induced signals along a long straight stem of the common sedge and got a distinct pattern of amplitude minima & maxima - a sinusoid shape, repeated regularly along the whole length. We speculate (in absence of a better explanation) that the pattern represents standing waves which originate from outgoing and rebound waves adding up, but no one has been able to prove that. So I thought I'd calculate the expected amplitude from known parameters at each point and compare it to the actual data. The attempt, of course, crashed on the way through my inability to do math/physics, and I wonder if someone may point me in the right direction, at least. I'm afraid nobody has done such a thing before, though, and if this is really the case, I'd really appreciate if someone can figure it out. We could probably even arrange a co-authorship of the article for the helpful contributor (but I can't promise that 100% right now).
Anyway, to the data:
We stimulated the stem artificially with a minishaker, using pure-tone 100 Hz vibration with the velocity of 10-3 m/s (I express it with velocity because it's more relevant for the system studied, but it can easily be converted to amplitude).
The stem is circular in cross-section, around 5 mm in diameter, and 1,65 m high. It was vibrated 1 cm below the top which was cut cleanly off. I also observed equivalent pattern in shorter stems.
We don't know exactly the velocity of propagation, the best approximation I have is 95 cm/s. I don't know about the elasticity of the stem either, but I can find out if necessary. Attenuation is around 0.3 dB/cm.
There are a couple additional assumptions we made, which may of course be wrong, but it's the best we can do:
- the mass loading of the stem due to it being fixed to the minishaker doesn't significantly affect the system more than 10 cm away
- the stem's cross section is uniform enough to simplify as completely uniform and/or irrelevant to the propagation of surface vibration
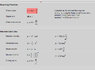
The first thing I tried was the basic formula for standing waves (the one here), but I got much shorter wavelength than observed experimentally. Unfortunately, I don't know whether the formula isn't good for this purpose or if the error was mine. That's what I can think of right now. Let me know if there's anything else of importance that I left out.
Thanks.
At my institution, we are researching communication of certain bugs, which use low-frequency, substrate-borne vibrations as signals. Those signals are so well tuned to the physical properties of plants the bugs live on, that they transmit with very little attenuation, and so they rebound (probably) several times from stem boundaries before dying down. As far as we know, those are surface vibrations, transmitted in the form of bending waves. I measured the amplitude of artificially-induced signals along a long straight stem of the common sedge and got a distinct pattern of amplitude minima & maxima - a sinusoid shape, repeated regularly along the whole length. We speculate (in absence of a better explanation) that the pattern represents standing waves which originate from outgoing and rebound waves adding up, but no one has been able to prove that. So I thought I'd calculate the expected amplitude from known parameters at each point and compare it to the actual data. The attempt, of course, crashed on the way through my inability to do math/physics, and I wonder if someone may point me in the right direction, at least. I'm afraid nobody has done such a thing before, though, and if this is really the case, I'd really appreciate if someone can figure it out. We could probably even arrange a co-authorship of the article for the helpful contributor (but I can't promise that 100% right now).
Anyway, to the data:
We stimulated the stem artificially with a minishaker, using pure-tone 100 Hz vibration with the velocity of 10-3 m/s (I express it with velocity because it's more relevant for the system studied, but it can easily be converted to amplitude).
The stem is circular in cross-section, around 5 mm in diameter, and 1,65 m high. It was vibrated 1 cm below the top which was cut cleanly off. I also observed equivalent pattern in shorter stems.
We don't know exactly the velocity of propagation, the best approximation I have is 95 cm/s. I don't know about the elasticity of the stem either, but I can find out if necessary. Attenuation is around 0.3 dB/cm.
There are a couple additional assumptions we made, which may of course be wrong, but it's the best we can do:
- the mass loading of the stem due to it being fixed to the minishaker doesn't significantly affect the system more than 10 cm away
- the stem's cross section is uniform enough to simplify as completely uniform and/or irrelevant to the propagation of surface vibration
The first thing I tried was the basic formula for standing waves (the one here), but I got much shorter wavelength than observed experimentally. Unfortunately, I don't know whether the formula isn't good for this purpose or if the error was mine. That's what I can think of right now. Let me know if there's anything else of importance that I left out.
Thanks.