Homework Help Overview
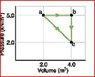
The discussion revolves around the change in internal energy for one mole of an ideal diatomic gas transitioning along a diagonal path in a thermodynamic process. Participants are exploring the relationship between internal energy, temperature, and the equations governing these concepts.
Discussion Character
- Exploratory, Conceptual clarification, Mathematical reasoning
Approaches and Questions Raised
- Participants discuss the equation for internal energy in relation to temperature and attempt to reconcile different paths taken in the process. Questions arise about the specific equation for internal energy and the necessary variables to calculate it.
Discussion Status
Some participants have offered guidance on using specific equations related to internal energy and work done. There is an acknowledgment of the need for temperature change and specific heat capacity in the calculations. Multiple interpretations of the problem are being explored, particularly regarding the paths taken in the process.
Contextual Notes
Participants are working within the constraints of a homework assignment, which may limit the information available or the methods they can employ. There is an emphasis on understanding the relationships between internal energy, heat, and work in the context of thermodynamics.