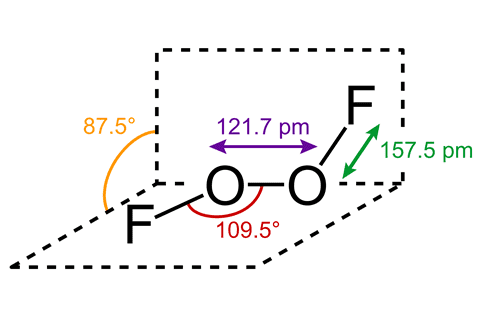
Discussion Overview
The discussion revolves around the structure of the chemical compounds FOOF and FOOOOF, exploring their stability, potential existence, and methods for determining their structures. The conversation includes references to literature and techniques relevant to the study of these compounds.
Discussion Character
- Exploratory
- Technical explanation
- Debate/contested
Main Points Raised
- Some participants mention that FOOF is known to decompose at approximately -160°C, raising questions about its stability at standard temperature and pressure (STP).
- There is a reference to the informal history of liquid rocket propellants, suggesting that compounds up to FOOOOOOF may exist, but without a clear structure provided.
- One participant suggests that determining the structure of these compounds might require extreme conditions or specialized techniques, such as matrix isolation spectroscopy.
- Another participant discusses the process of preparing the compound in situ with an inert gas like argon and using IR spectroscopy to deduce the structure, expressing uncertainty about the complexity of interpreting the resulting spectrum.
- There is mention of using computational methods, such as ab initio calculations with programs like Gaussian, to compare calculated IR spectra with observed data.
Areas of Agreement / Disagreement
Participants express varying levels of trust in the sources cited and the feasibility of working with these compounds. There is no consensus on the stability of FOOF or the methods for determining the structures of FOOF and FOOOOF, indicating multiple competing views and unresolved questions.
Contextual Notes
Limitations include the dependence on specific conditions for stability and the complexity of interpreting IR spectra for unusual compounds. The discussion reflects a range of assumptions about the properties and behaviors of these chemical species.