You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
What is Reaction: Definition and 1000 Discussions
A chemical reaction is a process that leads to the chemical transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive elements where both electronic and nuclear changes can occur.
The substance (or substances) initially involved in a chemical reaction are called reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more products, which usually have properties different from the reactants. Reactions often consist of a sequence of individual sub-steps, the so-called elementary reactions, and the information on the precise course of action is part of the reaction mechanism. Chemical reactions are described with chemical equations, which symbolically present the starting materials, end products, and sometimes intermediate products and reaction conditions.
Chemical reactions happen at a characteristic reaction rate at a given temperature and chemical concentration. Typically, reaction rates increase with increasing temperature because there is more thermal energy available to reach the activation energy necessary for breaking bonds between atoms.
Reactions may proceed in the forward or reverse direction until they go to completion or reach equilibrium. Reactions that proceed in the forward direction to approach equilibrium are often described as spontaneous, requiring no input of free energy to go forward. Non-spontaneous reactions require input of free energy to go forward (examples include charging a battery by applying an external electrical power source, or photosynthesis driven by absorption of electromagnetic radiation in the form of sunlight).
A reaction may be classified as redox in which oxidation and reduction occur or nonredox in which there is no oxidation and reduction occurring. Most simple redox reactions may be classified as combination, decomposition, or single displacement reactions.
Different chemical reactions are used during chemical synthesis in order to obtain a desired product. In biochemistry, a consecutive series of chemical reactions (where the product of one reaction is the reactant of the next reaction) form metabolic pathways. These reactions are often catalyzed by protein enzymes. Enzymes increase the rates of biochemical reactions, so that metabolic syntheses and decompositions impossible under ordinary conditions can occur at the temperatures and concentrations present within a cell.
The general concept of a chemical reaction has been extended to reactions between entities smaller than atoms, including nuclear reactions, radioactive decays, and reactions between elementary particles, as described by quantum field theory.
View More On Wikipedia.org
The substance (or substances) initially involved in a chemical reaction are called reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more products, which usually have properties different from the reactants. Reactions often consist of a sequence of individual sub-steps, the so-called elementary reactions, and the information on the precise course of action is part of the reaction mechanism. Chemical reactions are described with chemical equations, which symbolically present the starting materials, end products, and sometimes intermediate products and reaction conditions.
Chemical reactions happen at a characteristic reaction rate at a given temperature and chemical concentration. Typically, reaction rates increase with increasing temperature because there is more thermal energy available to reach the activation energy necessary for breaking bonds between atoms.
Reactions may proceed in the forward or reverse direction until they go to completion or reach equilibrium. Reactions that proceed in the forward direction to approach equilibrium are often described as spontaneous, requiring no input of free energy to go forward. Non-spontaneous reactions require input of free energy to go forward (examples include charging a battery by applying an external electrical power source, or photosynthesis driven by absorption of electromagnetic radiation in the form of sunlight).
A reaction may be classified as redox in which oxidation and reduction occur or nonredox in which there is no oxidation and reduction occurring. Most simple redox reactions may be classified as combination, decomposition, or single displacement reactions.
Different chemical reactions are used during chemical synthesis in order to obtain a desired product. In biochemistry, a consecutive series of chemical reactions (where the product of one reaction is the reactant of the next reaction) form metabolic pathways. These reactions are often catalyzed by protein enzymes. Enzymes increase the rates of biochemical reactions, so that metabolic syntheses and decompositions impossible under ordinary conditions can occur at the temperatures and concentrations present within a cell.
The general concept of a chemical reaction has been extended to reactions between entities smaller than atoms, including nuclear reactions, radioactive decays, and reactions between elementary particles, as described by quantum field theory.
View More On Wikipedia.org
-

Armature Reaction: Interpoles & Cross Magnetizing Effect
How do interpoles reduce armature reaction in dc machine? What is exactly the cross magnetizing effect ??- cnh1995
- Thread
-
- Tags
- Reaction
- Replies: 1
- Forum: Electrical Engineering
-

What is the correct solar neutrino reaction equation and how was it discovered?
Hi mfb: The following messages were in the now closed thread "Neutrinos-Antineutrinos in the universe". Bill_K said (msg #2): Most of the neutrinos we detect are solar neutrinos coming from p + P → d + e+ + ν. I said (msg #5): Is the "P" here a typo where "p" was intended? If not, what does...- Buzz Bloom
- Thread
- Replies: 21
- Forum: High Energy, Nuclear, Particle Physics
-
G
What happens if the normal reaction force is less than zero?
Homework Statement why the object will fall of when the normal reaction force is less than zero ? P/s : the normal reaction force is in downward direction... How if if the normal reaction force is less than zero ? the normal reaction is in upward direction ? Homework EquationsThe Attempt at a...- goldfish9776
- Thread
- Replies: 8
- Forum: Introductory Physics Homework Help
-
G
Direction of reaction force in a circular motion
Homework Statement Thewheel is rotated in the anticlockwise direction...The correct reaction force is drawn using the blue pen . I don't understand why the reaction force isn't directed to the left ? which is drawn using the red colour ? IMO, the red colour should be correct because the...- goldfish9776
- Thread
- Replies: 13
- Forum: Introductory Physics Homework Help
-
G
Direction of normal reaction of the Ferriw wheel at the high
Homework Statement As you can see , the writer state that the direction of normal reaction force(R) is acted upwards , I have another book state that the normal reaction force (R) is acted downwards , which one is correct ? Homework EquationsThe Attempt at a Solution [/B]- goldfish9776
- Thread
- Replies: 15
- Forum: Introductory Physics Homework Help
-
I
Help for calculation of the heat of the following reaction
Anybody can help for calculation of the heat of the following reaction? Some background: Ligand X bind to protein M, the product is MX. Binding affinity of X to M is K = [MX]/([M]*[X]). [MX], [M], [X] are the concentrations of each stuff after the reaction finished. The ratio of MX to total M...- Icecream
- Thread
-
- Tags
- Calculation Heat Reaction
- Replies: 5
- Forum: Biology and Chemistry Homework Help
-
U
What is the expected rate of reactions in the detector?
Homework Statement A reactor fires out ##10^{21}## neutrinos per second. A detector containing ##10m^3## of liquid which contains 30 carbon atoms every 60 hydrogen atoms. The detector-reactor distance is ##1000##. The cross section for the reaction is ##\sigma = 10^{-46} m^2##, and the density...- unscientific
- Thread
- Replies: 1
- Forum: Advanced Physics Homework Help
-

Would the Earth Have A Delay Reaction if the Sun disappeared
So, I heard the other day from someone online who claimed that if the sun would to suddenly disappear that the Earth would still travel in its regular path for 8 minutes before breaking off. The person claimed that because it takes light 8 minutes to reach Earth, and that since nothing can...- Dopplershift
- Thread
- Replies: 6
- Forum: Other Physics Topics
-
A question regarding action and reaction
I hit a bench with my fist, I experience equal and opposite reaction... Science says, it is because the nuclei of the atoms of my fist come close to the nuclei of atoms of the bench and experience repulsion. Am I on the right track?...Are there other explanations?- Deepak K Kapur
- Thread
- Replies: 11
- Forum: Other Physics Topics
-

Find Torque Caused by Beam Pulling Down on Another
Homework Statement Hi guys, I'm currently working on a homework question but I'm a little bit stuck although I do know what I'm meant to be doing. My question asks me to find the torque being caused by a beam pulling down on another. I'll explain a little more the context of it - there is one...- Alexander1
- Thread
-
- Tags
- Force Reaction Reaction force
- Replies: 18
- Forum: Introductory Physics Homework Help
-

Star Nuclear Reaction: P+P, D+P, He3 and CNO Cycle
Dear PF Forum, I'm just wondering about P+P reaction in star. Because of its pressure, in the core of the star, hydrogens fuse to become deuterium. Its the complete reaction P + P -> D D + P -> He3 He3 + He3 -> He3 + P + P and if the star is big enough, it can undergo CNO cycle, But what if...- Stephanus
- Thread
- Replies: 2
- Forum: Astronomy and Astrophysics
-

Why do double displacement reaction occur?
What's the reason for which double displacement reaction occur? Why there is a need for ion exchange between two compounds? They are already in a bonded state, a stabilised one. So what benefit would a double displacement reaction give? The ionic reaction AgNO3 +NaCl - - - - > AgCl(ppt) +...- Quotes
- Thread
-
- Tags
- Displacement Reaction
- Replies: 4
- Forum: Chemistry
-
S
What Is the Q-Value of Pu-239 Fission into Sr-92, He-4, and Neutrons?
Homework Statement Fission Reaction: Pu-239 + gamma --> Sr-92 + ? + 3n Q-Value = ? (Answer in MeV, correct to 5 significant figures) Atomic Masses (u) Alpha Particle = 4.00150618 He-4 = 4.0026032497 Pu-239 = 239.0521565 Sr-92 = 91.911030 n (neutron) = 1.00866501 Homework Equations...- says
- Thread
- Replies: 27
- Forum: Introductory Physics Homework Help
-
S
Direct energy conversion from p+Li-7 fusion reaction?
I have a question about direct energy conversion; if I want to directly extract electrical energy from the fusion reaction p+Li-7 reaction which generates He nuclei, with a kinetic energy of 8 MeV, what is the most suitable method of doing that? Should some method with charge separation be used...- Sven Andersson
- Thread
- Replies: 1
- Forum: Other Physics Topics
-
Y
Produced bubble in a chemical reaction
Hi, I have a very interesting but challenging question. Consider any chemical reaction that involves gas production (e.g. H2 gas is produced in KOH etching silicon). As we know, the produced gas will detach from the surface and reaction can continue. What if the gas doesn't detach from the...- Yinxiao Li
- Thread
- Replies: 1
- Forum: Materials and Chemical Engineering
-
T
Determination of specific heat in a reaction
Is there a way to determine the specific heat of a reaction before said reaction is introduced. thanks in advance for all of your inputs.- thisguy
- Thread
- Replies: 7
- Forum: Thermodynamics
-
A
State of matter in chemical reaction
Does anybody know where I could find worksheet to practice naming the state of matter in chemical reactions (with answers in the worksheet to check) as I have problems with that. Especially deciding when it is aqueous. I know (well at least i am right) that when you have acid as reactants it has... -

Reaction of thiols with vinyl ethers
Homework Statement Find the product: 2. Relevant concepts Thiols react with vinyl ethers according to anti-markovnikov's rule The Attempt at a Solution After reading Jerry March's book, i came to know that it follows free radical mechanism and that is why it gives anti markovnikov's...- AdityaDev
- Thread
- Replies: 4
- Forum: Biology and Chemistry Homework Help
-

Alkylation of Imines: achiral to chiral compounds
I found this in "March's Advanced organic chemistry" After that step, on using HCl in pentane, you will get 4-methyl-3-Heptanone. So this method was used to convert achiral 2-pentanone to a chiral compound, by first converting it to the compound on left by addition reaction and then using a 2... -

##\frac {t_{1/2}}{t_{3/4}}## for an nth order reaction
Homework Statement The question: For an nth order reaction ##\frac {t_{1/2}}{t_{3/4}}## depends on (n isn't equal to 1) : (A) initial concentration only (B) 'n' only (C) initial concentration and 'n' both (D) sometimes 'n' and sometimes initial concentration. Homework Equations...- mooncrater
- Thread
-
- Tags
- Reaction
- Replies: 5
- Forum: Biology and Chemistry Homework Help
-
W
Gamow peak and nuclear reaction rate
It's known that the nuclear reaction rate (inside a Star) can be determined with $$R_{ab}=n_a n_b\left<\sigma v\right> \, \approx \, n_a n_b \Big(\frac{8}{\pi m_e}\Big)^{1/2} \frac{S(E_0)}{(k_BT)^{3/2}} \Delta \frac{\sqrt{\pi}}{2} e^{-3E_0/k_BT},$$ where ##k_B## is Boltzmann constant, ##T##...- wieblinger
- Thread
- Replies: 2
- Forum: Astronomy and Astrophysics
-
R
Reaction of SOCl2 with White Phosphorus: Product Analysis
Homework Statement The product formed in the reaction of SOCl2 with white phosphorus is 1. PCl3 2. SO2Cl2 3. SCl2 4. POCl3 Homework Equations NA The Attempt at a Solution I can google that but I want to know that how can we know it intuitively or by ourselves? It was asked in a test and in...- Raghav Gupta
- Thread
- Replies: 11
- Forum: Biology and Chemistry Homework Help
-
J
Think the Mass is correct, unsure on Reaction Force
Homework Statement [/B]Homework Equations [/B] kg/m3 = mass / volume Downward forces on the column = Weight + Additional force (F=ma) The Attempt at a Solution [/B] kg/m3 = mass / volume Therefore, 2400 = mass / (0.3 x 0.4)...- jaja binks
- Thread
-
- Tags
- Force Mass Reaction Reaction force
- Replies: 8
- Forum: Engineering and Comp Sci Homework Help
-
A
Atmospheric Substitution Reaction
There is an ever constant increase of CO2 and CH4 in the atmospheric, thanks to global warming and greenhouse gases. I came across this equation while looking over changing gas compositions in the atmosphere. CO2 + CH4 ----> 2H2O + C2 Can someone explain me how I can figure out the amount of... -
G
Quantum Physics: The Reaction of a Photon Passing Through a Slit in a Plate
We got a light source and the light comes through a slit in a plate. A photon passes through the slit and ends up somewhere on a screen not in the straight direction due to diffraction. The question is, in quantum physics, does the plate feel the reaction of the photon deflection (1) at the time...- goran d
- Thread
-
- Tags
- Diffraction Reaction
- Replies: 2
- Forum: Quantum Physics
-
N
Got Gallium, what reaction is this?
I know little to nothing about chemistry, but am fascinated by its physics. Bought some gallium off amazon, which came shipped via AIR from china! (my God I hope that wasn't a passenger plane) and wow is it ever neat stuff. Any ways of course I wanted to try putting some Gallium on Aluminum... -

Direction of equilibrium if you remove heat from exothermic reaction
If you remove heat from an exothermic reaction will the equilibrium shift to the products or to the reactants according to le chatelier's principle- lonely_nucleus
- Thread
- Replies: 13
- Forum: Chemistry
-
S
Trigger for a Zn oxidation reaction in a battery Reference
Who is the trigger of the oxidation reaction. Copper which attracts electrons from the Zinc or solution which makes Zinc losing electrons and become positively charged. I am struggling to understand what actually makes Zinc to enter the solution and leave 2 electrons behind. If there is no wire... -
S
Trigger for a Zn oxidation reaction in a battery
Who is the trigger of the oxidation reaction. Copper which attracts electrons from the Zinc or solution which makes Zinc losing electrons and become positively charged. I am struggling to understand what actually makes Zinc to enter the solution and leave 2 electrons behind. If there is no wire...- Slava
- Thread
- Replies: 9
- Forum: Other Physics Topics
-

Is 3FeCl2 to 2FeCl3 and Fe(s) a Disproportionation Redox Reaction?
Homework Statement Determine the oxidizing agent, reducing agent, element oxidized, element reduced, # of electrons lost per atom, and # of electrons gained per atom. State if the reaction is not redox. 3FeCl2(aq) ------> 2FeCl3(aq) + Fe(s) Homework EquationsThe Attempt at a Solution...- Elvis 123456789
- Thread
- Replies: 1
- Forum: Biology and Chemistry Homework Help
-
H
State of matter by reversing reaction
Homework Statement Imagine a hypothetical process in which the methane molecule, CH4, is "expanded," by simultaneously extending all four C−H bonds to infinity. We then have the process: CH4 (g) → C(g) + 4H(g) Compare the reaction for the "expansion" of methane with the reverse of the reaction...- henry3369
- Thread
- Replies: 1
- Forum: Biology and Chemistry Homework Help
-
J
Reaction forces on simple 2 strut truss
Homework Statement Calculate reaction forces R1x, R1y, R2x, R2y. Load of 100N. Node 1 and 2 are fixed. Distances listed on image. Homework Equations [/B] I have: ∑Fx = 0 = R1x + R2x ∑Fy = 100 = R1y + R2y ∑M2 = 0 = (-100 x 0.1) + R1 x 0.06 + R1y x 0.02 ∑M1 = 0 = (-100 x 0.04) + (-R2x x...- James C
- Thread
- Replies: 6
- Forum: Engineering and Comp Sci Homework Help
-

Energy balance -- Physical interpretation of Q
Hello PF! I have a simple question on energy balances. Suppose you have an isothermal reactor where you want to have an exothermic reaction happening at 450 °C. The simplified energy balance for the reactor is Q = ΔH. Supposing ΔH = -150 kcal, then Q = -150 kcal. Does this mean that, a) in order...- MexChemE
- Thread
- Replies: 4
- Forum: Materials and Chemical Engineering
-
C
Firefigting nozzle pressure and reaction
A ladder pipe with a 2" tip at 80 psi nozzle pressure will have approximately ____ pounds of nozzle reaction. Now I've been out of college and out of practice with my math and physics skills for quite a while. With studying my firefighting materials for operations, a lot of the statements by... -
T
Torque Reaction Force of Transmission and Drivetrain
Hello, As I understand it there is an equal and opposite reaction for every mechanical action. In the case of a vehicle drivetrain the engine generates a torque at the flywheel or flex plate which becomes an input to the transmission and ultimately the wheels. The rollong resistance of the...- thender
- Thread
- Replies: 10
- Forum: Mechanical Engineering
-

Calculating ΔH°rxn for a Reaction
Homework Statement Given the data in the table below, ΔH°rxn for the reaction 4NH3 (g) + 5O2 (g) → 4NO (g) + 6H2O (l) is ________ kJ. Substance ΔH∘f(kJ/mol) H2O (l) -286 NO (g) 90 NO2 (g) 34 HNO3 (aq) -207 NH3 (g) -46 Homework Equations ΔH°rxn = ΔH°products -...- Drakkith
- Thread
-
- Tags
- Reaction
- Replies: 4
- Forum: Biology and Chemistry Homework Help
-
E
Will Ca(HCO3)2(aq) + 2NH3(aq) react?
Hi, I'm wondering if an aqueous solution of Calcium Bicarbonate and Ammonia would react to form Ammonium Bicarbonate and Calcium Hydroxide. Additionally, would Calcium Carbonate react with Ammonia in solution to form Ammonium Carbonate? EJ -
S
How does enthelpy change as the reaction proceeds?
My concept of enthalpy change is that the change is based on limits that approach zero. Something like the heat evolved/absorbed per second (but instead of second its a infinitely small). A+B→C ΔH=100J/mole of A (standard enthalpy change) And as such when the reaction first proceeds the... -
C
Enthelpy of reaction calculation?
Limestone stalactites and stalagmites are formed in caves by the following reaction: Ca2+(aq) + 2HCO3-(aq) --> CaCO3(s) + CO2(g) + H2O(l) . If one mol of CaCO3 forms at 298 K under 1 atm pressure, the reaction performs 2.48 kJ of P - V work, pushing back the atmosphere as the gaseous CO2 forms...- Centralscience
- Thread
- Replies: 6
- Forum: Biology and Chemistry Homework Help
-
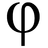
Linear reaction force on motors?
I have two motors(m1 &m2) placed on a vertical position(on the ##y-axis##), that have gears connected to their shafts, and I placed the gears next to one another. Fixing them to move together. I now powered both motors allowing their torques to oppose one another and cancel out each other's...- PhiowPhi
- Thread
- Replies: 8
- Forum: Mechanical Engineering
-

Product formed by the reaction
Please see the attached picture. I'm not sure which hydrogen would be anti-periplanar. Any help would be appreciated. Thanks!- 8008jsmith
- Thread
- Replies: 1
- Forum: Biology and Chemistry Homework Help
-
J
Statics problem involving moments and reaction forces
Homework Statement The right angle boom which supports the 230-kg cylinder is supported by three cables and a ball-and-socket joint at O attached to the vertical x-y surface. Determine the reactions at O and the cable tensions. Homework Equations M [/B]= r x F unit vector =...- June W.
- Thread
- Replies: 4
- Forum: Engineering and Comp Sci Homework Help
-

Pyrite roasting -- Mass balance with chemical reaction
Homework Statement A certain pyrite ore contains 85% of FeS2 and 15% of inerts. This ore is introduced into a roasting furnace with 20% excess air, in order to oxidize the FeS2 in the reaction: \textrm{FeS}_2 + \frac{11}{4}\textrm{O}_2 \rightarrow \frac{1}{2}\textrm{Fe}_2 \textrm{O}_3 +...- MexChemE
- Thread
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-
H
Why Is Krypton the Product in This Nuclear Reaction?
Can someone please explain to me why the answer to the following question is Krypton? A step by step solution would be ideal please! (b)The following reaction occurs in a nuclear reactor: 235U+01n →141Ba+X+301n+202.5MeV (i) Identify the element X. Many thanks! (Question is Q12 b part i...- Hooly
- Thread
- Replies: 5
- Forum: Advanced Physics Homework Help
-
A
Moment reaction with rotation and offset CG
Hey folks, I created and verified a model with hand calculations, and decided to re-use my same logic.. Which now is giving me false numbers. Was wondering if you guys could help me out. Setup/Verification (See attached Reaction_Forces picture): Body in motion being held down by straps... -
S
The Initial Reaction of a Voltaic Cell
I just want to start by saying that I have found many questions very similar to this one using the search bar, but I still don't understand exactly what happens initially in a simple voltaic cell with a salt bridge (Daniel cell). For example, I am trying to figure out what initially happens in... -
D
Why Does C6H5CH(Cl)CH3 < (CH3)3 CCl < C6H5C(CH3)2 Cl Undergo SN1 Reaction?
Homework Statement i was told that the the ascending order of the compound undergo SN1 reaction is C6H5CH(Cl)CH3 < (CH3)3 CCl < C6H5C(CH3)2 Cl... it's a past year question . I am sure tha ans is correct. Can someone explain why? Generally , the most reactive is the compound (CH3)3 CCl , Am i...- desmond iking
- Thread
-
- Tags
- Reaction
- Replies: 1
- Forum: Biology and Chemistry Homework Help
-
Reaction Mg (s) with NaCl+H2O2+KOH solution
Homework Statement I just want to know what reaction occurs when magnesium is inserted into NaCl+H2O2+KOH solution. Homework Equations What happened with each component in that solution ? I would be very grateful if you could help me. The Attempt at a Solution- y@_zzz
- Thread
-
- Tags
- Reaction
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-
Conflicting descriptions of Alka-Seltzer reaction?
Alka seltzer contains 1916 mg of NaHCO3 and 1000 mg of citric acid. According to some descriptions of the reaction between alka-seltzer and water and then HCl (in stomach), there is an excess of NaHCO3 in the alka-seltzer in relation to the citric acid and the benefits from alka-seltzer come...- david lewine
- Thread
- Replies: 6
- Forum: Chemistry
-
J
Am I reading this reaction right?
In the late 19th early 20th century, sulfuric acid production was a huge deal, and even today, conversion of SO2 to SO3 (contact process) is something that people take great pains to optimize. But I also see references here on physics forums as well as elsewhere which assert that sulfur dioxide...- JeffEvarts
- Thread
- Replies: 8
- Forum: Chemistry