rikku95
- 1
- 0
- TL;DR
- Heat Transfer of an Deware Nitrogen Container
Hello guys, 
So like the Titels says I am Trying to get an Approximation of the Heat Transfer which ocours in a Deware container. Unfortunately all my calculations so far seem to suggest way to low Boil off rates for my Container its just to efficient, here is a summary of my Attempts.
First of all I got myself the Heat Conduction formula for an Cylindrical Objekt and for a Circle and my heat conduction coefficient.


After i calculated the Heat Conduction in Watts,the next step was the Latent Heat of LN2 And the Density of LN2 which is it ;

So the Boil off of LN2 form -196 to 21C° in watts is
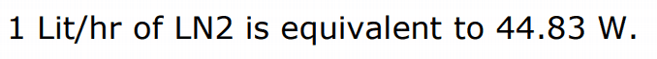 So that´´ s it, after i plug my numbers in i always get way too low Boil off rates for my containers.What did i do wrong?What did I forget?
So that´´ s it, after i plug my numbers in i always get way too low Boil off rates for my containers.What did i do wrong?What did I forget?
Heres an Example
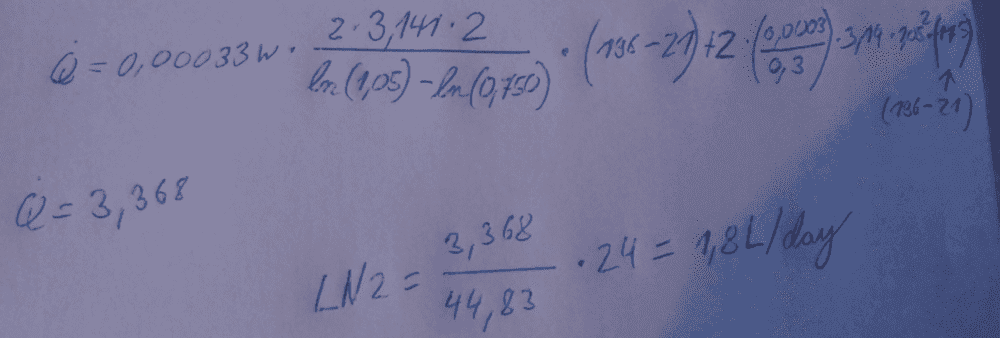
These are Meters yes, so at an outer radius of 1,05 m and an height of 2 meters only Produces 1,8L/day, i don't think so.
Please help me i am in Great pain
So like the Titels says I am Trying to get an Approximation of the Heat Transfer which ocours in a Deware container. Unfortunately all my calculations so far seem to suggest way to low Boil off rates for my Container its just to efficient, here is a summary of my Attempts.
First of all I got myself the Heat Conduction formula for an Cylindrical Objekt and for a Circle and my heat conduction coefficient.
After i calculated the Heat Conduction in Watts,the next step was the Latent Heat of LN2 And the Density of LN2 which is it ;
So the Boil off of LN2 form -196 to 21C° in watts is
Heres an Example
These are Meters yes, so at an outer radius of 1,05 m and an height of 2 meters only Produces 1,8L/day, i don't think so.
Please help me i am in Great pain
