Discussion Overview
The discussion revolves around the effects of atmospheric pressure on measurements in a Torricelli barometer, particularly in relation to Pascal's law. Participants explore the principles of pressure in fluids, the definition of Pascal's law, and the implications for understanding barometric measurements.
Discussion Character
- Technical explanation
- Conceptual clarification
- Debate/contested
Main Points Raised
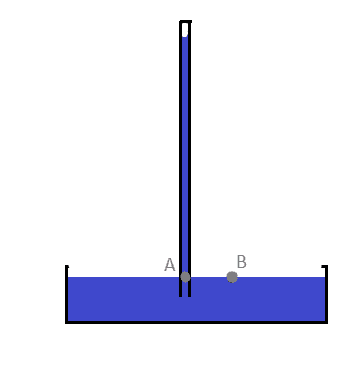
- One participant questions why atmospheric pressure is not included in the pressure calculation at point A in the barometer, suggesting that it should be considered according to Pascal's law.
- Another participant asserts that the tube is closed and contains a vacuum at the top, indicating that atmospheric pressure acts on the liquid at point B, causing it to rise at point A due to Pascal's law.
- A participant clarifies that the barometer measures the unknown atmospheric pressure and references a Wikipedia article for further information.
- There is a discussion about a common misconception regarding Pascal's law, with one participant stating that it describes pressure acting isotropically at a specific point, rather than uniformly throughout the fluid.
- A participant expresses surprise at the previous comment and questions the conditions under which pressure is transmitted uniformly in a fluid, seeking clarification on the relationship to Pascal's law.
- Another participant notes that pressure varies with depth in a gravitational field, indicating that uniform pressure transmission occurs only in static equilibrium without gravity.
- One participant mentions that the pressure at point A should include both the hydrostatic pressure from the mercury column and the saturated vapor pressure of mercury above it, referring to the space above the mercury as a 'Torricelli vacuum.'
Areas of Agreement / Disagreement
Participants express differing views on the application of Pascal's law and the role of atmospheric pressure in the barometer's operation. There is no consensus on the correct interpretation of these principles, and multiple competing views remain.
Contextual Notes
Participants highlight limitations in textbook definitions and the need for clarity regarding the conditions under which Pascal's law applies. The discussion also touches on the implications of gravitational effects on pressure in fluids.