Discussion Overview
The discussion revolves around the number of halogens removed in E2 reactions, focusing on the conditions necessary for elimination and the role of various reagents in substitution and elimination processes. Participants explore theoretical aspects, mechanisms, and specific examples related to E2 reactions and substitution reactions.
Discussion Character
- Homework-related
- Exploratory
- Debate/contested
- Technical explanation
Main Points Raised
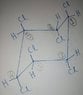
- One participant questions the clarity of the problem statement and discusses the requirement for anti-periplanar geometry for elimination to occur.
- Another participant suggests that stereoinversion may be necessary to achieve the required geometry for E2 reactions, noting the use of "halogens" instead of specifying "chlorines."
- There is a proposal to consider substitution with PBr3, with questions about whether the reaction would stop at mono-substitution.
- Some participants express uncertainty about the outcomes of using PBr3, suggesting it may lead to a mixture of products and emphasizing the need for substitution before elimination.
- Questions arise regarding the conditions under which NaI and acetone lead to substitution versus elimination, with requests for specific examples.
- Concerns are raised about the likelihood of elimination occurring with NaI alone, with discussions about the conditions that might favor elimination.
- One participant speculates about the possibility of a Markovnikov elimination yielding trichlorobenzene, while others note the importance of the spatial arrangement of groups in E2 mechanisms.
- There is a suggestion that the reactions should be treated as separate cases, with a focus on the number of halogens removed in the combined E2 reactions.
Areas of Agreement / Disagreement
Participants express differing views on the mechanisms and outcomes of the reactions discussed, with no consensus reached on the specifics of how many halogens will be removed or the exact nature of the reactions involved.
Contextual Notes
Participants highlight the importance of spatial arrangements in E2 mechanisms and the potential for multiple products due to varying reaction pathways. There is also mention of the need for additional reagents to facilitate certain reactions, indicating limitations in the assumptions made.