krizam
- 1
- 0
For some of my work, I am investigating methods of scanning for nuclear material. As part of my study, I have run across Gamma Ray Spectroscopy: http://en.wikipedia.org/wiki/Gamma_spectroscopy"
What I am having trouble with is reading a gamma ray spectrum (I am a software engineer with a limited physics background).
I have two examples that are confusing me (from that wikipedia article):
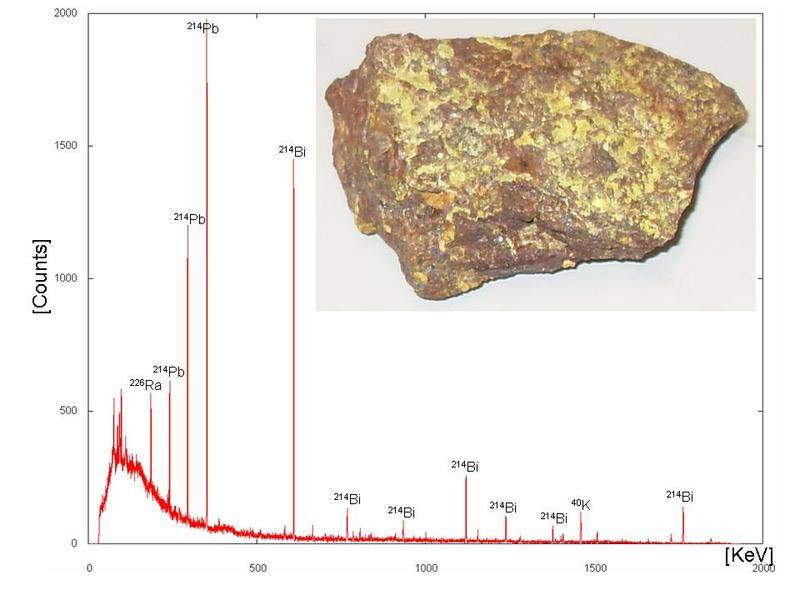
What do the axes actually mean? I get the concept of the "count" being the activity of the detector. What are the peaks showing? Do peaks at certain spots signify a "fingerprint" for an isotope (Such as 214 BI having 7 peaks?)?
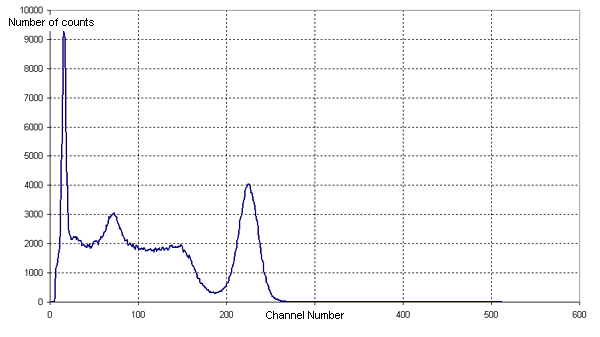
Why is the bottom axis on this graph labeled as "channel number"? How is that different then the previous graph? In the article it says for this figure that "An example of a NaI spectrum is the gamma spectrum of the cesium isotope 137Cs—see Figure XXX. 137Cs emits a single gamma line of 662 keV." Is this represented somehow in this figure?
Thanks for any help. I just am looking for a basic understanding so I can read these spectrums.
What I am having trouble with is reading a gamma ray spectrum (I am a software engineer with a limited physics background).
I have two examples that are confusing me (from that wikipedia article):
What do the axes actually mean? I get the concept of the "count" being the activity of the detector. What are the peaks showing? Do peaks at certain spots signify a "fingerprint" for an isotope (Such as 214 BI having 7 peaks?)?
Why is the bottom axis on this graph labeled as "channel number"? How is that different then the previous graph? In the article it says for this figure that "An example of a NaI spectrum is the gamma spectrum of the cesium isotope 137Cs—see Figure XXX. 137Cs emits a single gamma line of 662 keV." Is this represented somehow in this figure?
Thanks for any help. I just am looking for a basic understanding so I can read these spectrums.
Last edited by a moderator: