Tom79Tom
- 45
- 3
I have trouble understanding why we classify an inviscid adiabatic incompressible flow along a streamline as isentropic
I understand this from a Thermodynamic definition/explanation
$$dS = dQ/T$$
Adiabatic Invsicid
$$dQ =0= dS$$
So no heat added or lost no change in entropy I'm fine with that
From a Boltzmann definition I am less clear how is it possible that we can have additional gradients within in the volume and more ordered momentum yet have no change in available entropy
$$S=k_BlnW$$If we looked at a two different control volumes a straight pipe and a venturi separating a pressure difference
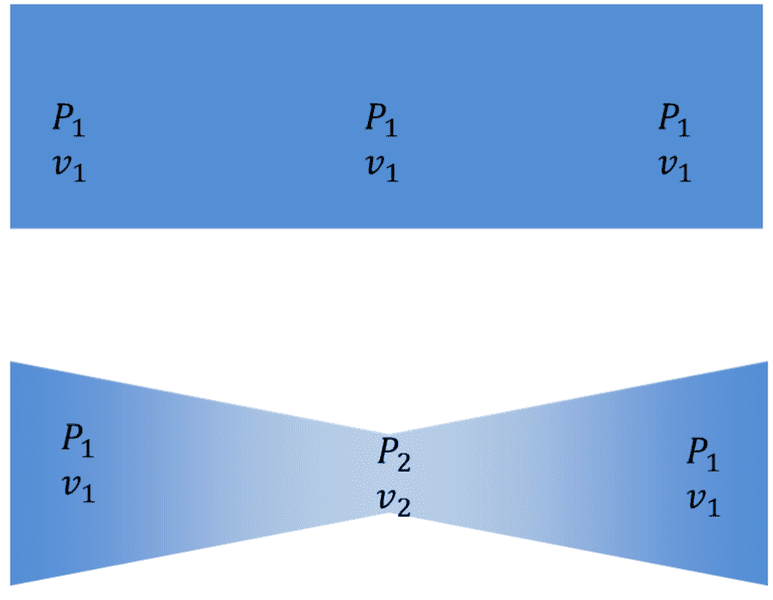
In an inviscid adiabatic flow, Starting pressure P1 and final pressure P1 are the same and equal , the flow rate Q in and out of the volume are the same but the presence of the Venturi causes additional gradients P1 to P2 and an increase in ordered kinetic energy from v1 to v2To me follow that this is a reduction in available microstates ∑{x,y,z,px,py,pz} compared to an inviscid flow without a venturi.
Understanding that no flow is not in thermodynamic equilibrium and that within each example flow the sum of microstates is constant so Entropy within the flow is constant how can we say that these flows have the same available microstates and entropy ?
Wouldn't it be better to say that it is a reversible isochoric process between differing macrostates along the flow ?
[1]: https://i.stack.imgur.com/N9lfB.png
I understand this from a Thermodynamic definition/explanation
$$dS = dQ/T$$
Adiabatic Invsicid
$$dQ =0= dS$$
So no heat added or lost no change in entropy I'm fine with that
From a Boltzmann definition I am less clear how is it possible that we can have additional gradients within in the volume and more ordered momentum yet have no change in available entropy
$$S=k_BlnW$$If we looked at a two different control volumes a straight pipe and a venturi separating a pressure difference
In an inviscid adiabatic flow, Starting pressure P1 and final pressure P1 are the same and equal , the flow rate Q in and out of the volume are the same but the presence of the Venturi causes additional gradients P1 to P2 and an increase in ordered kinetic energy from v1 to v2To me follow that this is a reduction in available microstates ∑{x,y,z,px,py,pz} compared to an inviscid flow without a venturi.
Understanding that no flow is not in thermodynamic equilibrium and that within each example flow the sum of microstates is constant so Entropy within the flow is constant how can we say that these flows have the same available microstates and entropy ?
Wouldn't it be better to say that it is a reversible isochoric process between differing macrostates along the flow ?
[1]: https://i.stack.imgur.com/N9lfB.png