DumpmeAdrenaline
- 80
- 2
Thread moved from the technical forums to the schoolwork forums
Note: I am self-studying Material and Energy balance courses and I haven't done fluid mechanics yet.
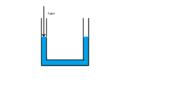
Case 1) Consider the manometer in the figure below. Levels on both sides of the manometer which are open to the atmosphere are equal.
If we analyse the forces acting on the left side of the manometer. We have the atmospheric force exerting a downward force on the fluid and since the fluid is not undergoing acceleration it is either moving at a non zero speed or is stationary. Therefore the fluid must be exerting an equal but upward force on the atmosphere.
Case 2) If we hook up the left side of the manometer to a gas source containing higher kinetic energy molecules than the air molecules on the right side exposed to the atmosphere then those gas molecules are exerting a higher pressure. What would happen in this case?
Will the gas molecules push the liquid, bounce, pass through the liquid as bubbles?
I wrote case (1) mainly to link the upward force the fluid exerts and what is it a function of?
Case 1) Consider the manometer in the figure below. Levels on both sides of the manometer which are open to the atmosphere are equal.
If we analyse the forces acting on the left side of the manometer. We have the atmospheric force exerting a downward force on the fluid and since the fluid is not undergoing acceleration it is either moving at a non zero speed or is stationary. Therefore the fluid must be exerting an equal but upward force on the atmosphere.
Case 2) If we hook up the left side of the manometer to a gas source containing higher kinetic energy molecules than the air molecules on the right side exposed to the atmosphere then those gas molecules are exerting a higher pressure. What would happen in this case?
Will the gas molecules push the liquid, bounce, pass through the liquid as bubbles?
I wrote case (1) mainly to link the upward force the fluid exerts and what is it a function of?