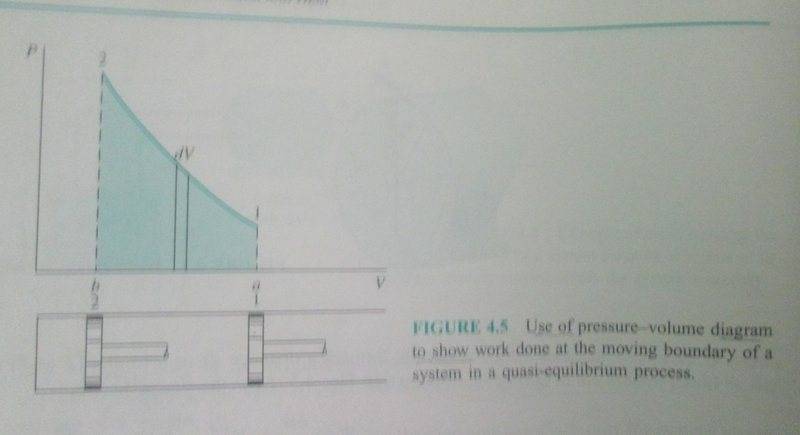
Discussion Overview
The discussion revolves around the characteristics of pressure-volume (P-V) diagrams for gas compression in a closed system, particularly focusing on whether the graph must always be non-linear and the implications of different temperature conditions on the shape of the curve.
Discussion Character
- Exploratory, Technical explanation, Debate/contested
Main Points Raised
- One participant questions whether the P-V graph must always be non-linear during gas compression, particularly under isothermal conditions, and raises the possibility of a horizontal graph if temperature decreases.
- Another participant suggests that the Ideal Gas Law (PV = NRT) should be assumed unless a different equation is specified, which could help in expressing P as a function of V and calculating work done.
- A repeated point emphasizes the need to focus on the shape of the curve rather than just the calculation of work done.
- One participant proposes that the compression process may be polytropic, introducing the equation PV^n = C, which adds to the confusion regarding the curve's shape.
Areas of Agreement / Disagreement
Participants express differing views on the shape of the P-V curve during gas compression, with no consensus reached on whether it must always be non-linear or if horizontal graphs are possible under certain conditions.
Contextual Notes
Participants have not resolved the implications of temperature changes on the P-V diagram shape, and there is ambiguity regarding the application of the Ideal Gas Law versus polytropic processes.