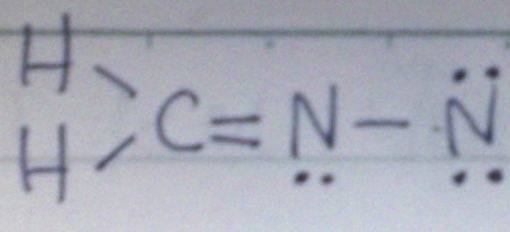Discussion Overview
The discussion revolves around the challenges of drawing Lewis structures for molecules containing nitrogen, specifically focusing on the example of H2CNN. Participants explore the correct bonding arrangements and the implications of valence bond structures.
Discussion Character
- Exploratory, Technical explanation, Homework-related
Main Points Raised
- One participant expresses difficulty in drawing the correct Lewis structure for H2CNN, initially proposing a double bond between carbon and nitrogen and a single bond between the nitrogens.
- Another participant suggests that while the initial structure is a valid representation, it may not be the most significant one due to the rightmost nitrogen having only a sextet, proposing a transformation to fulfill the octet rule for all atoms.
- A third participant requests additional examples of similar problems to practice, indicating a desire for further exploration of the topic.
- One participant acknowledges the help received, indicating that the discussion has been beneficial.
Areas of Agreement / Disagreement
Participants do not reach a consensus on the best Lewis structure for H2CNN, and multiple viewpoints regarding the significance of different bonding arrangements remain present.
Contextual Notes
There are limitations regarding the completeness of examples and the specific criteria for determining the importance of various Lewis structures, which are not fully resolved in the discussion.
Who May Find This Useful
Students and individuals interested in chemistry, particularly those learning about molecular structures and bonding involving nitrogen.