You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
What is Heat: Definition and 1000 Discussions
In thermodynamics, heat is energy in transfer to or from a thermodynamic system, by mechanisms other than thermodynamic work or transfer of matter. The various mechanisms of energy transfer that define heat are stated in the next section of this article.
Like thermodynamic work, heat transfer is a process involving more than one system, not a property of any one system. In thermodynamics, energy transferred as heat contributes to change in the system's cardinal energy variable of state, for example its internal energy, or for example its enthalpy. This is to be distinguished from the ordinary language conception of heat as a property of an isolated system.
The quantity of energy transferred as heat in a process is the amount of transferred energy excluding any thermodynamic work that was done and any energy contained in matter transferred. For the precise definition of heat, it is necessary that it occur by a path that does not include transfer of matter.Though not immediately by the definition, but in special kinds of process, quantity of energy transferred as heat can be measured by its effect on the states of interacting bodies. For example, respectively in special circumstances, heat transfer can be measured by the amount of ice melted, or by change in temperature of a body in the surroundings of the system. Such methods are called calorimetry.
The conventional symbol used to represent the amount of heat transferred in a thermodynamic process is Q. As an amount of energy (being transferred), the SI unit of heat is the joule (J).
View More On Wikipedia.org
Like thermodynamic work, heat transfer is a process involving more than one system, not a property of any one system. In thermodynamics, energy transferred as heat contributes to change in the system's cardinal energy variable of state, for example its internal energy, or for example its enthalpy. This is to be distinguished from the ordinary language conception of heat as a property of an isolated system.
The quantity of energy transferred as heat in a process is the amount of transferred energy excluding any thermodynamic work that was done and any energy contained in matter transferred. For the precise definition of heat, it is necessary that it occur by a path that does not include transfer of matter.Though not immediately by the definition, but in special kinds of process, quantity of energy transferred as heat can be measured by its effect on the states of interacting bodies. For example, respectively in special circumstances, heat transfer can be measured by the amount of ice melted, or by change in temperature of a body in the surroundings of the system. Such methods are called calorimetry.
The conventional symbol used to represent the amount of heat transferred in a thermodynamic process is Q. As an amount of energy (being transferred), the SI unit of heat is the joule (J).
View More On Wikipedia.org
-
T
Heat Transfer - Critical Radius of Insulation
A steel pipe with foam insulation is embedded in a concrete wall. The steel pipe is carrying cold water and therefore gains heat from outside atmosphere. Heat transfers through the concrete wall, foam insulation and then to the pipe. Is it possible to calculate critical radius of insulation for...- Thevan
- Thread
- Replies: 15
- Forum: Thermodynamics
-
E
I Calculating the Area of an RN Event Horizon with Specific Heat Formula
By definition ##C = T_H \dfrac{\partial S}{\partial T_H} \bigg{)}_Q## so given ##A=4S## we first need to work out the area of the event horizon. More specifically, let ##\Sigma## be a partial Cauchy surface of constant ##v## in ingoing EF ##(v,r,\theta, \phi)## co-ordinates then ##A## is the...- etotheipi
- Thread
-
- Tags
- Heat Specific Specific heat
- Replies: 1
- Forum: Special and General Relativity
-

Heat Transfer Formulae for Charcoal-Concrete Experiment
My experiment procedure is that I am using burning charcoal as the heat source, and there are three thermocouples used, first one measuring the temperature of the charcoal, second one measuring the inner temperature of the concrete block, third thermocouple is placed on the top part which is...- ahbin98
- Thread
-
- Tags
- Formulae Heat Heat transfer
- Replies: 1
- Forum: Mechanical Engineering
-

Engineering Average heat transfer coefficient (forced convection)
So firstly, I don't understand if the mass flow rate is for steam or for water. If it is for water, I know I can find the heat transfer rate using equation:Q=mcdeltaT. But then I don't know how to find h (the average heat transfer coefficient) because I don't know the surface area (As). I can...- bardia sepehrnia
- Thread
- Replies: 6
- Forum: Engineering and Comp Sci Homework Help
-
M
What are the key parameters affecting heat transfer in thin tubes?
Hi, I am trying to understand how I can estimate the time it takes for a fluid at room temperature flowing through a thin capillary glass tube (2 mm ID) connected to an oven to reach the equilibrium temperature (oven temperature). Assuming the oven is preheated and the tube inside the oven is...- MSM
- Thread
-
- Tags
- Fluid flow Heat Heat transfer
- Replies: 15
- Forum: General Engineering
-

Vaporization latent heat vs boiling point
answer is 78oCdelta G = delta H - T delta S -235,310- (-277,690) = -38.56e3- T (282.59-160.70) T = -664K I am not sure if my concept is correct. May anyone help a little bit on that please? thank you- yecko
- Thread
- Replies: 2
- Forum: Introductory Physics Homework Help
-
L
Engineering Heat transfer from composite pipe -- Is my answer right?
Im practicing the questions in the problem book and seem to be getting different answers to the book can somebody check cheers. [Answers: 57.99 W/m: 1739.7 W: 84.9ºC] textbook answers A water pipe of bore 65 mm bore and 6mm wall thickness, carrying water at 85ºC is insulated with one layer of...- lee123456789
- Thread
-
- Tags
- Composite Heat Heat transfer Pipe
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-
E
A query about heat capacity and specific heat capacity
Homework Statement:: why does heat capacity depend on the mass/size of the object when it's units is J/K , and why is specific heat capacity dependent on the material/substance when it's unit is J/kgK? Relevant Equations:: Q=Cθ Q=mcθ -- ellieee
- Thread
- Replies: 8
- Forum: Thermodynamics
-
E
Specific heat capacity & heat capacity
the answer is 1/2 , but when I did the working out, I got 2/1.. so I'm quite confused- ellieee
- Thread
- Replies: 6
- Forum: Introductory Physics Homework Help
-
K
Rate of reaction? (UV and heat)
I remember when I was in high school, heat was very often said to be a factor of speeding up chemical reactions that we all know, because heat is actually energy, energy will excite the molecules and make them easier to collide with other molecules or leave its own molecules (breaking bond)... -
A
Calculating specific heat capacity from entropy
Hey guys! I'm currently struggling with a specific thermodynamics problem. I'm given the entropy of a system (where ##A## is a constant with fitting physical units): $$S(U,V,N)=A(UVN)^{1/3}$$I'm asked to calculate the specific heat capacity at constant pressure ##C_p## and at constant volume...- approx12
- Thread
- Replies: 1
- Forum: Advanced Physics Homework Help
-

What's the deal with plasma physicists setting heat cap. ratio to 3?
This has persistently bugged me in my intro plasma course. They keep using ##\gamma = 3## aka ##N_d = 1## (where ##N_d## is the number of degrees of freedom in the molecule) as an approximation. See for example, the Bohm-Gross dispersion curve. I can tell you from deriving this that the factor...- Twigg
- Thread
-
- Tags
- Heat Physicists Plasma Ratio
- Replies: 2
- Forum: Thermodynamics
-
J
Relative Humidity and Temperature Changes when adding Heat
Can anyone help me with this? -How much heat must be added to 3.5 m3 /s of moist air with a dry bulb temperature of 10°C and a relative humidity of 60% to raise the temperature of the air by 17°C? • What will be the relative humidity of the air once this heat is added? • What is the power...- john ryan
- Thread
- Replies: 11
- Forum: Introductory Physics Homework Help
-
J
Calculating how much heat must be added to increase temp
Can anyone help me with this? -How much heat must be added to 3.5 m3 /s of moist air with a dry bulb temperature of 10°C and a relative humidity of 60% to raise the temperature of the air by 17°C? • What will be the relative humidity of the air once this heat is added? • What is the power...- john ryan
- Thread
- Replies: 1
- Forum: Thermodynamics
-
U
Heat conduction from an isotherm spherical cap
On the surface of a semi-infinite solid, a point heat source releases a power ##q##; apart from this, the surface of the solid is adiabatic. The heat melts the solid so that a molten pool forms and grows. Let's hypothesize that the pool temperature is homogeneously equal to the melting...- umby
- Thread
- Replies: 20
- Forum: Thermodynamics
-
Thermodynamics: Internal Energy, Heat and Work Problem
Well, internal energy is the sum of the kinetic and potential energies of all the molecules within a given mass of a substance; this energy is associated with the random, disordered motion of the molecules. An example of internal energy is compressed gases; since gases occupy the total volume...- AN630078
- Thread
- Replies: 1
- Forum: Introductory Physics Homework Help
-

Can heat flow into a body without increasing the mean kinetic energy?
Yes, heat can flow into a body without increasing the mean kinetic energy of its molecules. Transferring heat energy to an object will raise its internal energy, this will not necessarily cause an increase in temperture. Specific latent heat is the energy required to change the state of one...- AN630078
- Thread
- Replies: 1
- Forum: Introductory Physics Homework Help
-
M
Heat pump efficiency calculations - Compressor power issue
The heat pump comprises of the 4 components: evaporator, compressor, condenser and expansion valve. Thermal power required to heat the building: 12.1 kW at condensing temperature tc = 44.3 deg C For the evaporator: vaporizing temperature tv = -7 deg C Subcooling temperature for the heat pump Δ...- Martin Harris
- Thread
- Replies: 23
- Forum: Materials and Chemical Engineering
-

Verify this function is a solution of the heat equation
I spent hours looking at this and cannot figure out where the error is. I'm wondering if there is an error before the boxed expression. @Orodruin and @PeroK may I ask for your assistance?Consider a solution ##u:[0,\infty)\times \mathbb{R}^n\rightarrow \mathbb{R}## of the heat equation, ie...- docnet
- Thread
-
- Tags
- Function Heat Heat equation
- Replies: 2
- Forum: Calculus and Beyond Homework Help
-
J
Calculating the waste heat of Carnot engine
The answer is 6470 J. So since I have the two temperatures I could calculate the efficiency. First I convert to kelvin then get an efficiency of 0.35481. Now I can use e=W/Qin to get Qin. I get a value of 10033.54J. Now I can use e=(Qin-Qout)/Qout to get Qout, the waste heat. I get 7405.9 J...- JoeyBob
- Thread
-
- Tags
- Carnot Carnot engine Engine Heat
- Replies: 4
- Forum: Introductory Physics Homework Help
-

Solving the heat equation in 1D
(3) To solve the initial value problem $$\begin{cases} \partial_t\phi-\partial^2_x\phi=0 & \text{in}\quad (0,\infty)\times R \\ \phi(0,\cdot)=\psi & \text{on}\quad \{t=0\}\times R \end{cases}$$ we use the fundamental solution in 1D $$\Phi_1(t,x)=\frac{1}{\sqrt{4\pi...- docnet
- Thread
-
- Tags
- 1d Heat Heat equation
- Replies: 3
- Forum: Calculus and Beyond Homework Help
-
J
Efficiency of heat engine question
So efficiency is W/Qin. W= 0 for isochoric processes and for the isobaric, P(change in V). So W=Pi(Vi-Vf)+Pf(Vi-Vf) Qin is negative Qs.This would happen at step 2 and 3. For the isobaric, Q=ncv(change in T) and for isochoric, Q=ncp(change in T). Now if I put everything in the equation I get...- JoeyBob
- Thread
-
- Tags
- Efficiency Engine Heat Heat engine
- Replies: 14
- Forum: Introductory Physics Homework Help
-
S
HVAC Attic to Pool Heat Exchange, will car radiator collect heat?
My attic gets very hot even with the exhaust fan I installed. I would like to move than heat to my swimming pool. I currently have a FAFCO solar pool heater where the pool pump pumps water up onto my roof and thru the 12' long solar panel and down back into the pool. I would like to free up roof...- shanngrila
- Thread
- Replies: 6
- Forum: DIY Projects
-
M
Specific latent heat of fusion of ice question
a) Ew = EL + Ei mwc(T2-T1) = miL + mic(T2-0) 160 x 4.2 x (30 - 20) = 20L + (20 x 4.2 x 20) L = (6720 - 1680)/20 = 252 J/g b) accounting for 3 gram of melted ice 160 x 4.2 x (30 - 20) = (20 - 3) + (20 x 4.2 x 20)...- maxim07
- Thread
-
- Tags
- Fusion Heat Ice Latent heat Specific
- Replies: 9
- Forum: Introductory Physics Homework Help
-
P
Rate of heat flux from from hot gas in a pipe to water outside
I have solved the first 2 parts. For the 3rd part, I have obtained the equation: T(x) - T0 = (T1 - T0)e^(-Φx/fc), where f = fm in the question. How do I obtain that expression for H? Thank you!- phantomvommand
- Thread
- Replies: 2
- Forum: Introductory Physics Homework Help
-
J
Find rate of temperature change using heat capacity, density and area
So first I found rate of heat change using the above equation, with T=883K, e=1, SA= 6*l^2=21.66 Now dQ/dt=746593.71 W Now I am not sure entirely what to do next. They give density so I likely have to get the mass from that, M=pV,=1.9^3*4037=27689.783 kg. My issue is that I don't know how to...- JoeyBob
- Thread
- Replies: 4
- Forum: Introductory Physics Homework Help
-
T
MTW Exercise 22.7 -- Calculate the law of local energy conservation for a viscous fluid with heat flow
I've come to a grinding halt with this and I can't see a way forward. Can someone please take a look at what I've done so far and let me know if what I have done is OK and then if it is, give me a hint on how to proceed. First up, Is ## u \cdot \nabla \cdot T = u_\alpha...- TerryW
- Thread
- Replies: 2
- Forum: Advanced Physics Homework Help
-
S
Calculate energy required to heat water using a steam coil
I'm trying to complete this past exam paper Q. Water volume = 200hl = 20000L 1L=10^-3 m^3 20000L = 200 m^3 Density of water at 15 deg C = 999 kg m^3 Density = Mass/Volume 999 kg m^3 = Mass/(200 m^3) Mass of water = (200)(999) = 199800 kg Heat required to to heat 199800 kg water: Q =m C dT =... -

A Solution of nonhomogeneous heat equation problem
This is from Evans page 50. I'm sure it's something simple, but I don't follow the change from $$ \frac{\partial}{\partial t} \quad \text{to} \quad -\frac{\partial}{\partial s}$$ and from $$ \Delta_x \quad \text{to} \quad \Delta_y$$. \begin{gather*} \begin{split} u_t(x,t) - \Delta u(x,t) & =...- Shackleford
- Thread
- Replies: 6
- Forum: Differential Equations
-

Heat transfer in thermodynamics
In answering questions (a.) Why heat intake in this system is ## \Delta Q_{sys} = ( \Delta Q_{hot water} + \Delta Q_{cold water} ) / 2 ## where. ##\Delta Q_{hot water} = c(T_1 - T_f) ## ##\Delta Q_{cold water} = c(T_2 - T_f) ## I think T or T_f should be between T_1 and T_2 But why is the...- Another
- Thread
- Replies: 9
- Forum: Introductory Physics Homework Help
-
D
Chemistry Thermochemistry and the heat of complete combustion
The balanced reaction wil be : ##2C_6H_{6(l)}+15O_{2(g)}->12CO_{2(g)}+6H_2O_(l)## in order to compute the the standard enthalpy of reaction : ##\Delta H°_{f} H_2O_(l)= -285,8 \frac {KJ}{mol}##; ##\Delta H°_{f} CO_{2(g)}= -393,5 \frac {KJ}{mol}##; ##\Delta H°_{f} C_6H_{6(l)}=49,04 \frac...- DottZakapa
- Thread
- Replies: 4
- Forum: Biology and Chemistry Homework Help
-
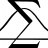
Boiling Water using Heat vs. Vacuum
If you bring a pot of water to boil, you will notice that the bubbles form on the bottom surface of the pot. This seems to make sense intuitively as this is the hottest surface, so this would be where the boiling action starts. When I was in school during a thermo lab we pulled a vacuum in a... -
K
Help solving this Heat Equation please
I want to solve the heat equation below: I don't understand where the expression for ##2/R\cdot\int_0^R q\cdot sin(k_nr)\cdot r \, dr## came from. The r dependent function is calculated as ##sin(k_nr)/r## not ##sin(k_nr)\cdot r##. I don't even know if ##sin(k_nr)/r## are orthogonal for...- Karl Karlsson
- Thread
- Replies: 3
- Forum: Calculus and Beyond Homework Help
-

Net heat in a thermodynamic cycle
In AB, dU=0 (Isothermal) Hence dQ=dW W1= RT ln(2V/V) = RT ln(2) = 800R*ln(2) Q1=W1 = 800R*ln(2) Q2= Cp(300-800)=-500Cp = -500*2.5R Q3 = Cv(800-300) = 500Cv = 500*1.5R So Q= 800R*ln(2) -500*2.5R + 500*1.5R = 800R*ln(2) -500R Hence net heat absorbed is 800R*ln(2) -500R But. Suppose I find...- Kaguro
- Thread
-
- Tags
- Cycle Heat Net Thermodynamic
- Replies: 2
- Forum: Introductory Physics Homework Help
-
S
What is the preheat temperature for the water entering the gas boiler?
Summary:: I need to know the preheat temperature of the water entering the gas boiler (which i think is the outlet temperature of the cold water Tc,o leaving the heat exchanger) Hi, I'm doing a design project of a hairpin heat exchanger. The problem is: In a public shower, the city water is...- Seta
- Thread
-
- Tags
- Design Heat Heat exchanger
- Replies: 10
- Forum: Engineering and Comp Sci Homework Help
-

Steam Speed from a Heat Ruptured 120 Gallon Water Tank?
PROBLEM: A 120 gallon tank (26" diameter, 60" height) containing 40 Gallons of water is heated up until the tank ruptures from overpressure at 125 PSI (maximum tank rating). How to calculate the initial escaped steam velocity at the ruptured tank, assuming the gash is 2 feet long in the...- seeker11
- Thread
- Replies: 24
- Forum: Mechanical Engineering
-

Efficiency of a heat engine given a P-V diagram of its process
I post the working in the attached file- XSummer32
- Thread
- Replies: 1
- Forum: Introductory Physics Homework Help
-
P
Heat Diffusion Equation - Using BCs to model as an orthonormal system
I've tried to show b) by using the sine Fourier series on ##[0,2a]##, to get ##g_k = \Sigma_{n=0}^{2a} \sqrt\frac{2}{a} Sin(q_k x)## Therefore ##\sqrt\frac{2}{a} = \frac{1}{a} \int_0^{2a} Sin(q_kx)g_k dx## These are equal therefore it is an orthonomal basis. I'm not sure if this is correct so...- physconomic
- Thread
- Replies: 2
- Forum: Advanced Physics Homework Help
-
R
Engineering Heat Capacity and the Coefficient of thermal expansion
thanks!- ReuvenD10
- Thread
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-

Does Copper Conduct Heat Better than cPVC?
Hi Folks, I'm 70, so I often confuse myself! Ha ! My question is: Do materials both absorb and release heat equally? So when I evaluate different materials for thermo efficiency, does a copper pipe's surface absorb solar heat at the same rate it gives off heat? My question is specific to a...- Steven Bolgiano
- Thread
- Replies: 2
- Forum: Thermodynamics
-

Two-phase flow heat transfer coefficient
Hi, I'm looking to find a very very basic model to calculate the heat transfer coefficient in a tube with given heat power exchanged ##Q##. The fluid inside the tube starts sub-cooled (##T_{in}## is known) but exits the tube with a quality of steam around 0.15 (pressure is supposed constant)...- dRic2
- Thread
- Replies: 34
- Forum: General Engineering
-

Heat Engine Problem - Get Help Now
Please help answering- mavidias
- Thread
-
- Tags
- Engine Heat Heat engine
- Replies: 1
- Forum: Introductory Physics Homework Help
-
M
How can I calculate the heat transfer in an air to air heat exchanger?
I am considering building this type of heat exchanger, as I have seen several youtube videos on the build. The inside tube is aluminum, I am planning on building one from the top of my roof to nearly the floor, so ill say 5 feet. I am thinking of using a 1.5" inch aluminum tube, and bringing in...- mess
- Thread
-
- Tags
- Air Heat Heat exchanger
- Replies: 22
- Forum: Mechanical Engineering
-
K
I Heat Wave Equations: Explaining Delta t & x Approach to Zero
When the delta t and x approached zero, assumably it results in the whole fraction to be zero. The slide shows it will be ##k^2##. Can someone explain this to me, please? P.S. I have tried to search this up, I could not find anything related to the confusion.- Kajan thana
- Thread
- Replies: 3
- Forum: General Math
-
J
Debye Approximation of Heat Capacity in 1D
So really i am just unsure how to answer the last part of the question. I am unsure how to apply the low and high temperature limits the way i have done it. Do i set upper/lower limits on the integral and solve? If so i am not sure what to put Here is what he book has for 3d- jkthejetplane
- Thread
- Replies: 1
- Forum: Advanced Physics Homework Help
-
J
Heat related problem -- Mercury solidifying liberates some energy
I have used the heat equation and am not able to get the initial heat. This is what I have so far: 81060 = 7 * 140 * (change in temp) - (7 * 11300) + 7 * 140 ( change in temp) I found the change in temp to be 81.71K. But that does not help me with the initial temperature!- JennyLee1989
- Thread
- Replies: 15
- Forum: Introductory Physics Homework Help
-

What factors affect heat rejection in vacuum pump systems?
I'm working on a project to replace a couple of vacuum pump skids for a lab campus. The existing pumps are water-cooled, liquid-ring pumps. Since they are water cooled, there is effectively no heat rejection into the mechanical room(s) except through un-insulated exhaust pipes. The new pumps...- russ_watters
- Thread
- Replies: 3
- Forum: Mechanical Engineering
-

Heat transfer problem for a long rectangular bar
Summary:: Determine the temperature distribution in a bar (very long– 2D) with rectangular cross section, in steady state, with imposed flux at one face, convection at the opposing face (Tinf, h), and imposed temperature (T1) at the two remaining walls. I am trying to find the analytical...- Nusselt
- Thread
- Replies: 4
- Forum: Engineering and Comp Sci Homework Help
-
Gravity, heat, flooding, and centrifugal force on a tidal-locked planet
Hi. I'm a science fiction author whose first novel Hemispheres, published through RockHill Publishing, explored a tidal-locked planet (Gliese 581g) where a group of activists increased its rotation to bring daylight cycles to both hemispheres. Now I'm writing the sequel, and there's ecological...- Mark Everglade
- Thread
- Replies: 5
- Forum: Sci-Fi Writing and World Building
-
M
Optimizing Air Flow for CO2 and Heat Loss in Vans
My goal is to determine the ideal amount of air flow, in order to keep CO2 below 1000ppm and to minimize heat loss. I calculated the following based off a similar post in this forum, and I am hoping this can be verified so I know that i am in the right ballpark and going in the right direction...