JessicaHelena
- 188
- 3
Homework Statement
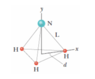
In the ammonia (NH3) molecule of the figure, the three hydrogen (H) atoms form an equilateral triangle, with the center of the triangle at distance d = 9.40 ✕ 10^−11 m from each hydrogen atom. The nitrogen (N) atom is at the apex of a pyramid, with the three hydrogen atoms forming the base. The nitrogen-to-hydrogen atomic mass ratio is 13.9, and the nitrogen-to-hydrogen distance is L = 10.14 ✕ 10^−11 m.
(a) What is the x coordinate of the molecule's center of mass?
(b) What is the y coordinate of the molecule's center of mass?
Homework Equations
x_cm = sum of (m*x)/M
(same for y)
The Attempt at a Solution
(a) I'm not quite sure how to deal with the two hydrogens towards the negative x-axis — they're not exactly on the axis at all.
(b) None of the H's have any y values, so I did 13.9*sqrt(L^2-d^2)/(13.9+3*1) where 3*1 accounted for the masses of the three H's, but apparently that answer is wrong, and I've no idea why it is. (I got 1.137*10^-(10).)