Homework Help Overview
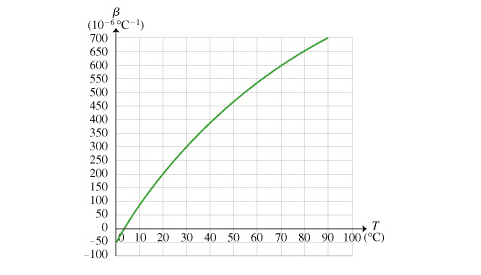
The discussion revolves around the density of water at specific temperatures, particularly focusing on the relationship between temperature changes and density variations. Participants are examining a graph that illustrates these changes and are trying to identify at what temperature water has the same density as at 1°C, as well as where the maximum density occurs.
Discussion Character
- Exploratory, Conceptual clarification, Mathematical reasoning, Assumption checking
Approaches and Questions Raised
- Participants are analyzing a graph to determine the temperature at which water's density is equal to that at 1°C and questioning the implications of the graph's behavior around 0°C and 4°C. There is also discussion about the significance of the slope and the concept of density changes with temperature.
Discussion Status
Some participants have provided insights into the graph's interpretation and the mathematical concepts involved, while others are seeking clarification on specific terms and the relationship between temperature and density. There appears to be a productive exploration of the topic, with various interpretations being discussed.
Contextual Notes
Participants mention the difficulty in reading the graph and the need for familiarity with calculus to fully understand the implications of the density changes. There is also a reference to the specific temperature at which water's density is highest, which is noted as 4°C.