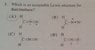Discussion Overview
The discussion centers around identifying an acceptable Lewis structure for Diazomethane, focusing on the resonance structures and the application of the octet rule. The scope includes homework-related inquiries and conceptual clarifications regarding formal charges and bonding in nitrogen.
Discussion Character
- Homework-related, Conceptual clarification, Debate/contested
Main Points Raised
- One participant asserts that structures C and D are incorrect, suggesting that structure A is preferable due to nitrogen's electronegativity, but later claims that the correct answer is actually structure B.
- Another participant prompts a check on the electron count around nitrogen in structure A.
- A follow-up response indicates that structure A has 4 bonds and 1 lone pair on nitrogen, questioning the validity of this configuration based on the octet rule.
- A link to the Wikipedia page on the octet rule is provided, possibly for further reference on the topic.
Areas of Agreement / Disagreement
Participants express differing views on the correctness of the Lewis structures, with no consensus reached on which structure is acceptable. The discussion remains unresolved regarding the validity of structure A and its implications for nitrogen bonding.
Contextual Notes
There are limitations regarding the assumptions made about nitrogen's bonding capabilities and the definitions of acceptable Lewis structures, which are not fully explored in the discussion.