JHUK
- 5
- 0
I posted in picture format to post on another website, but haven't found a reply yet:
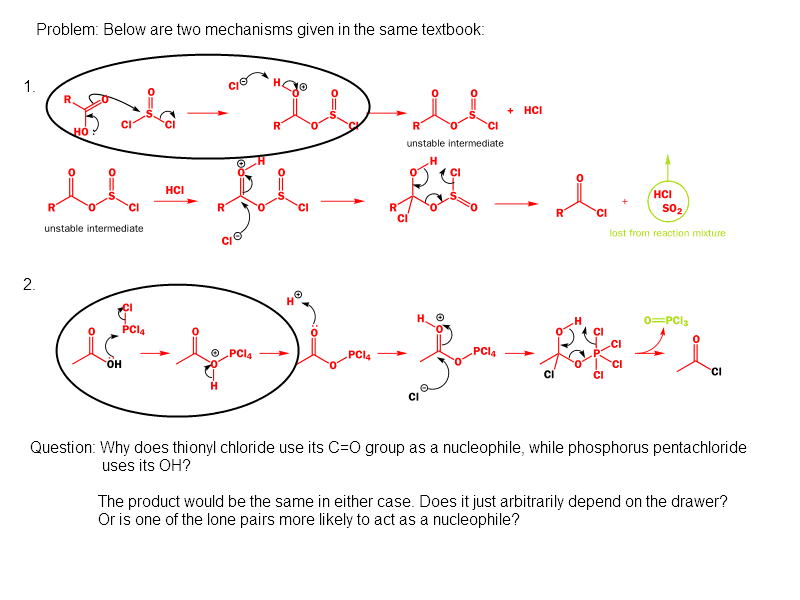
The discussion revolves around the mechanisms of nucleophilic attack involving oxygen atoms in carboxylic acid derivatives, particularly focusing on the roles of different oxygen atoms in the context of resonance structures and protonation states. The scope includes theoretical considerations and mechanistic insights.
Participants express differing views on the clarity of the question and the mechanisms involved, indicating that multiple competing interpretations remain without a consensus on the correct approach.
There are limitations in the clarity of the original question and the assumptions regarding the equivalence of oxygen atoms in different protonation states, which may affect the interpretations of the mechanisms discussed.
Yanick said:You're question is a little unclear because the mechanism shows nucleophilic attack by a -COOH group. The oxygens in these groups are actually equivalent and are best represented by resonance structures where the hydroxyl and carbonyl oxygens exchange.
sjb-2812 said:No, they are equivalent in the carboxylate anion, but not in the protonated version. Resonance structures do not involve the movement of protons.