ida
- 5
- 0
Hello everyone,
I have an rather large piston-cylinder containing gas that is compressed when the piston extends. Using three sensors I am measuring the pressure, temperature and volume of the gas while I force the piston out using a winch. The system is closed and no gas is released or added to the system while doing the experiments.
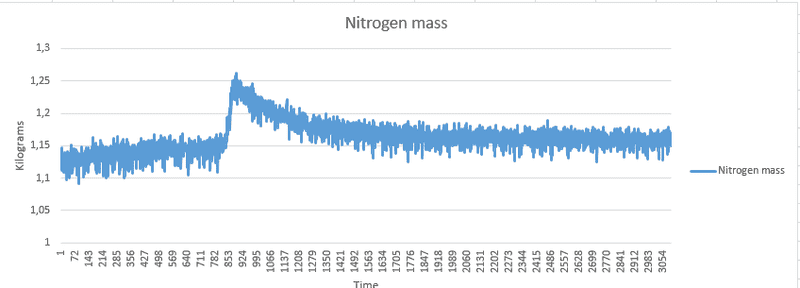
From the data collected I am calculating the gas-mass using the ideal gas law (PV=mRsT). I was expecting the gas-mass to be more or less constant, however the results show that the mass seems to change during the gas-compression phase, and slowly crawl back to the initially calculated mass from before the gas-compression phase was started.
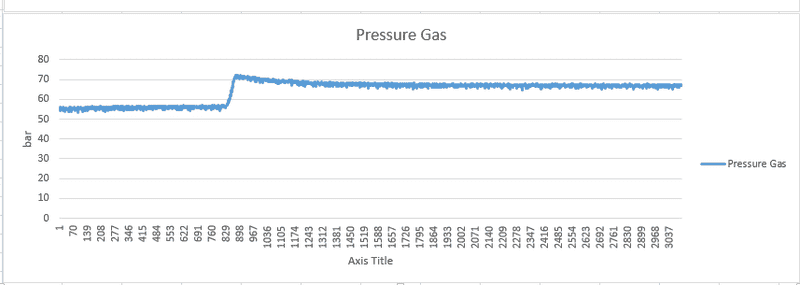
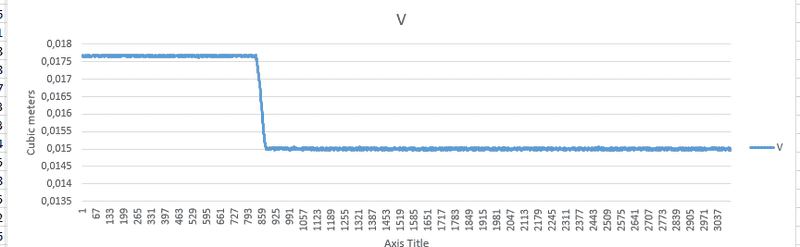
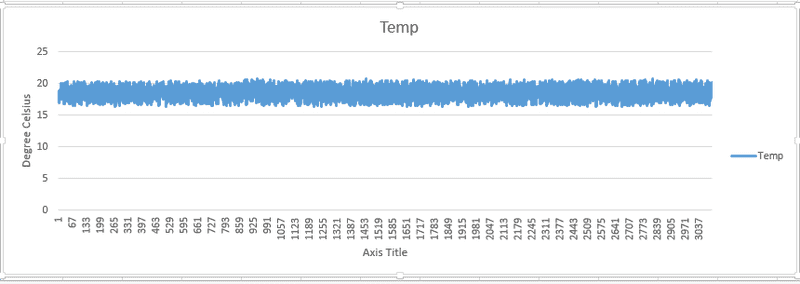
I've attached the state variables that i logged during my experiment. I started with the piston fully retracted, then pulled it out using a winch and kept it there for the rest of the experiment.
Could someone, with a bit more brains than me, enlighten me as to why the mass is not constant, and maybe point me towards another approach to calculating the mass of the gas in the cylinder?
My own hunch tells me that it got something to do with the limitations of the ideal gas law, but my thermodynamics knowledge is not sufficient to fully understand it. :)
I have an rather large piston-cylinder containing gas that is compressed when the piston extends. Using three sensors I am measuring the pressure, temperature and volume of the gas while I force the piston out using a winch. The system is closed and no gas is released or added to the system while doing the experiments.
From the data collected I am calculating the gas-mass using the ideal gas law (PV=mRsT). I was expecting the gas-mass to be more or less constant, however the results show that the mass seems to change during the gas-compression phase, and slowly crawl back to the initially calculated mass from before the gas-compression phase was started.
I've attached the state variables that i logged during my experiment. I started with the piston fully retracted, then pulled it out using a winch and kept it there for the rest of the experiment.
Could someone, with a bit more brains than me, enlighten me as to why the mass is not constant, and maybe point me towards another approach to calculating the mass of the gas in the cylinder?
My own hunch tells me that it got something to do with the limitations of the ideal gas law, but my thermodynamics knowledge is not sufficient to fully understand it. :)