twosockz
- 1
- 0
I am trying to experimentally determine the atomic scattering factors for NaCl using Laue photography. For an NaCl crystal, the intensity of the wave scattered at the unit cell is proportional to (4⋅(fCl+fNa))2 if h,k,l are even and to (4⋅(fCl-fNa))2 if h,k,l are odd. fNa and fCl are the atomic scattering factors which depend on sin(θ)/λ.
Therefore, I think that if sin(θ)/λ is approximately equal for two sets of h,k,l (one all even and one all odd), then we can combine the two equations to get:
Ieven+Iodd∝32⋅(fNa2+fCl2)
&
Ieven-Iodd∝64⋅fNa⋅fCl
(Is this correct so far?)
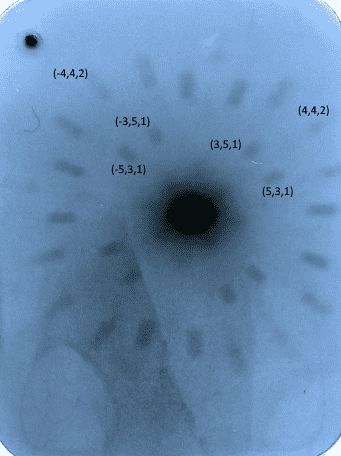
I plan to calculate the intensity ratios using pixel values of a scanned photographic x-ray film, as shown below. As such, we can then find the ratio of the scattering factors for Sodium and Chlorine using the equations above.
Attempting to use the (3,5,1) (odd) and (4,4,2) (even) Miller indices, which both have sin(θ)/λ ~ 0.05, I noticed a problem, as the second intensity equation implies that Ieven>Iodd, but, as shown below, this clearly isn't the case.
Are there any other factors affecting the intensity distribution that I am missing?

Therefore, I think that if sin(θ)/λ is approximately equal for two sets of h,k,l (one all even and one all odd), then we can combine the two equations to get:
Ieven+Iodd∝32⋅(fNa2+fCl2)
&
Ieven-Iodd∝64⋅fNa⋅fCl
(Is this correct so far?)
I plan to calculate the intensity ratios using pixel values of a scanned photographic x-ray film, as shown below. As such, we can then find the ratio of the scattering factors for Sodium and Chlorine using the equations above.
Attempting to use the (3,5,1) (odd) and (4,4,2) (even) Miller indices, which both have sin(θ)/λ ~ 0.05, I noticed a problem, as the second intensity equation implies that Ieven>Iodd, but, as shown below, this clearly isn't the case.
Are there any other factors affecting the intensity distribution that I am missing?

Attachments
Last edited: