How PET Scans Work: Positrons in Medical Imaging Explained
Table of Contents
Practical applications of fundamental theoretical physics
Positron Emission Tomography (PET)
Less often, people ask about potential applications of anti-matter. Both of those questions can be  answered with a common medical diagnostic technology, Positron Emission Tomography, or PET.
answered with a common medical diagnostic technology, Positron Emission Tomography, or PET.
Brief history of positrons
Positrons were first theorized by Paul Dirac in 1928 when he realized that his equation describing the electron admitted solutions for particles with positive as well as negative charges. They were observed four years later by Carl Anderson. Between the 1950s and the 1980s, the technology was developed to use positrons for medical diagnostics.
Meaning of “tomography”
The word tomography is often not heard outside of medical imaging, it refers to an image being reconstructed by adding together many individual slices. It is from the same Greek word as an atom, a-tomos being unsliceable.
How PET works
Producing the radioactive tracer
The radioactive isotope Fluorine-18 is produced in a particle accelerator by colliding a proton beam (with an energy of about a millionth that of the LHC) into water containing Oxygen-18. It is incorporated using chemistry in a glucose molecule called fludeoxyglucose (FDG). That is administered to a person, who is made of trillions of glucose combustion machines. As the glucose is spread through the body and metabolized, the fluorine decays (it has a half-life of just under two hours), emitting a positron. The positron quickly finds an electron, usually within two millimeters (there tend to be a lot of them inside things made of atoms), and annihilates, releasing two gamma photons with an energy of roughly 511 keV (the mass of an electron, times c##^2##). Because momentum is conserved, they fly off in opposite directions.
Detection and localization
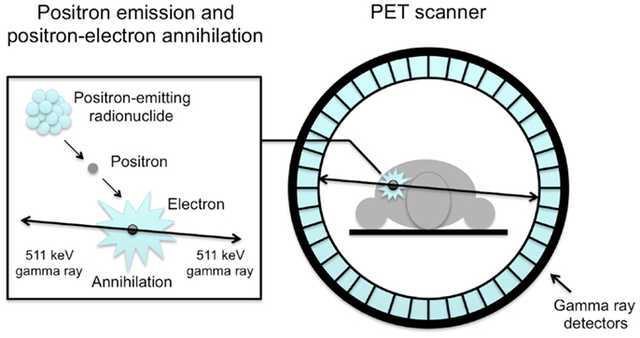 Now ideally, the person is lying stationary inside a ring of gamma detectors. The two photons are detected at two different locations around the ring, and the line connecting these tells you the axis on which they propagated. The difference in arrival time between the two photons tells you where along that axis they originated: light travels at roughly one foot per nanosecond, so if they originated six inches off-center, there would be a one-nanosecond delay between the two detections. This information lets you pinpoint where in the body the annihilation took place. In practice, time resolution may not be sufficient for this level of pinpointing, but multiple measurements of coincident photons allow for triangulation.
Now ideally, the person is lying stationary inside a ring of gamma detectors. The two photons are detected at two different locations around the ring, and the line connecting these tells you the axis on which they propagated. The difference in arrival time between the two photons tells you where along that axis they originated: light travels at roughly one foot per nanosecond, so if they originated six inches off-center, there would be a one-nanosecond delay between the two detections. This information lets you pinpoint where in the body the annihilation took place. In practice, time resolution may not be sufficient for this level of pinpointing, but multiple measurements of coincident photons allow for triangulation.
Image reconstruction and clinical use
Over time, enough events are detected to reconstruct an image. What we see when we look at a PET scan is essentially a map of where glucose is being metabolized in the body. The patient is relaxed in the scanner, so the muscles are not too active, but the brain is constantly churning. The bladder lights up because that’s where the stuff in our body tends to end up. But importantly, tumors are sites of high metabolism, and they light up under PET, and can be seen, diagnosed, and tracked.
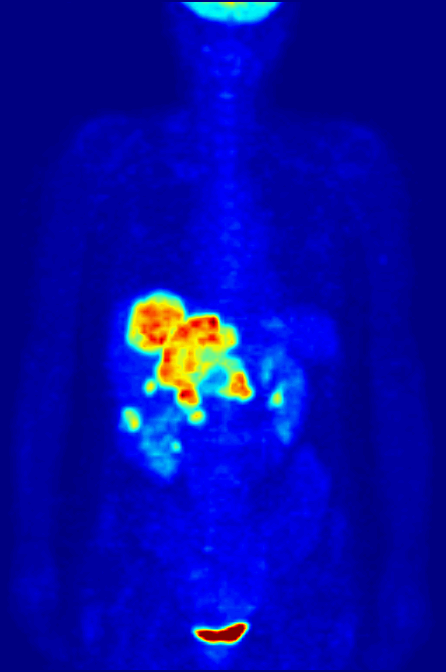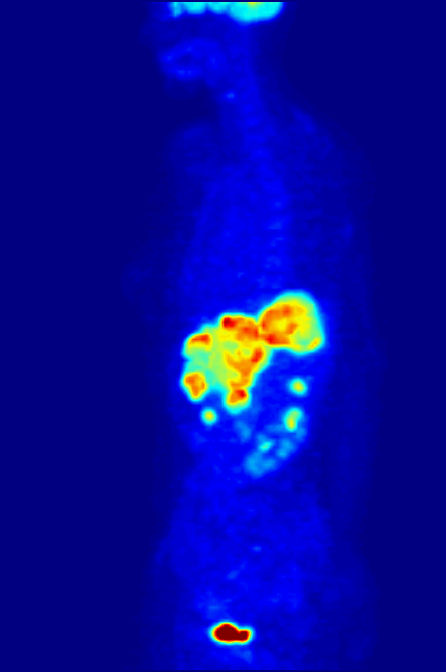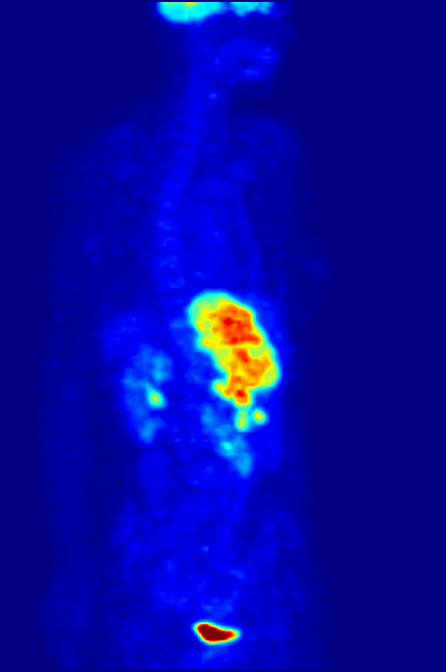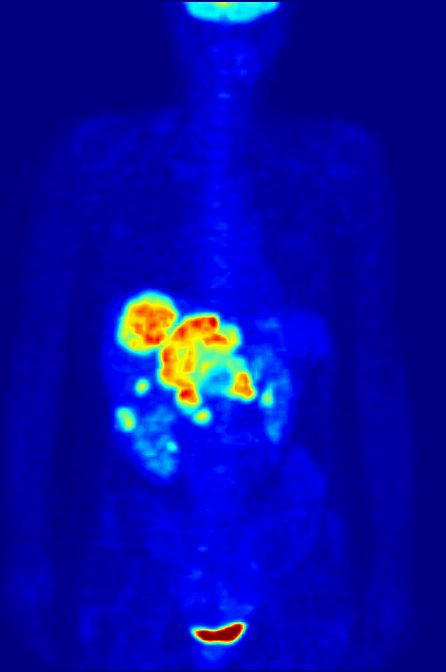
Broader implications of theoretical physics
What started as a theoretical physics investigation lead to a medical diagnostic technology for over half a century. Will the discovery of the Higgs boson or gravitational radiation lead to similar advances in technology? If I were a gambling man I would say no, but I don’t know what the future holds in store.
Ph.D. McGill University, 2015
My research is at the interface of biological physics and soft condensed matter. I am interested in using tools provided from biology to answer questions about the physics of soft materials. In the past I have investigated how DNA partitions itself into small spaces and how knots in DNA molecules move and untie. Moving forward, I will be investigating the physics of non-covalent chemical bonds using “DNA chainmail” and exploring non-equilibrium thermodynamics and fluid mechanics using protein gels.




“This is really cool.
What does it have to with Cerenkov radiation? How can Cerenkov radiation be created with gamma rays?”
It’s very difficult to detect gamma rays. One of its signature is that gamma rays tend to knock off electrons from atoms of the material, and these electrons are often so energetic, that it has a relativistic speed higher than the speed of light in that material. When this happens, you get Cerenkov radiation.
This is also the common technique to detect neutrinos. You don’t detect neutrinos, you detect the after effect of its collision with the water molecule resulting in a relativistic electron having speed higher than light in water (or whatever medium that is being used). This also creates a Cerenkov radiation.
Thus, the similarities between the two means that advancement in neutrino detection has a DIRECT effect and application in the detection technology for PET scans.
Zz.
Just left a presentation about brain imaging and they talked about PET. This researcher worked mainly on mapping memory in the brain. Seemed to prefer fMRI. Interesting stuff!
[ATTACH=full]97264[/ATTACH]
There are also other advances in physics beyond elementary particles that made this, and many other areas of medicine, possible. Note that there has to be a very good detector to detect the photons created. The area of detector and device physics is crucial here, because the process is only as good as what it can detect and display in the end. The sound coming from your hi-fi system is only as good as the speakers at the very end (or your headphones), no matter how much you spent on the rest of your sound system.
And interestingly enough, the drive to make better photodetectors comes out of basic, fundamental research. The need to have better detectors to detect the Cerenkov light made by neutrinos passing through water drives the technology to make better photodetectors, which in turn, [URL=’http://psec.uchicago.edu/other/Project_description_nobudgets.pdf’]trickles down to many other applications[/URL], including more efficient detectors for PET scans.
The moral of the story here is that, pick ANY medical device or procedure, and there is an extremely good chance that it came out of more than one area of physics, and that it benefited from the advances made not only in terms of physics knowledge, but also in terms of side-benefits of physics experiments.
Zz.
Always love to read about medical physics! It’s an important field!
This is really cool.What does it have to with Cerenkov radiation? How can Cerenkov radiation be created with gamma rays?
I worked on PET scanners for a while, and the typical detectors consist of Bismuth Germinate crystals glued onto photomultiplier tubes. The Bismuth Germinate crystals are transparent, and convert the impinging gamma rays (511KeV) to blue light (Cerenkov), which is detected by the photomultiplier tube and amplified to give an electrical pulse. These devices use Positronic Flourine as indictaced, but also Positronic Oxygen (half life 30 seconds) and other Positronic elements typically generated on-site using a low cost cyclotron.
Great summary!