Why Bother Teaching Mechanical Energy Conservation?
Note: It is assumed that the reader has read part I and part II of the series.
Table of Contents
Earlier parts
- Is Mechanical Energy Conservation Free of Ambiguity?
- Can We Do Better Than Mechanical Energy Conservation?
Preface
Because of what has already been said, there seem to be three options for proceeding with the teaching of mechanical energy conservation in introductory physics courses.
- Make no changes.
- Keep teaching mechanical energy conservation but fix it first.
- Abandon mechanical energy conservation and replace it with the law of energy conservation.
Option 1 is the default “do nothing” option; it is what we have now and we know exactly what it looks like. Option 3 has been presented in some detail in part II. Because option 2 has not been described so far, we will examine what it might look like before comparing them with each other. How the implementation of the last two options might be achieved is a question worth considering but not here. Instead, we will compare the pros and cons of each option. In the end, implementation notwithstanding, the reader may wish to consider the question “If you were asked to teach energy conservation, which option would you choose?”
Keep mechanical energy conservation but fix it
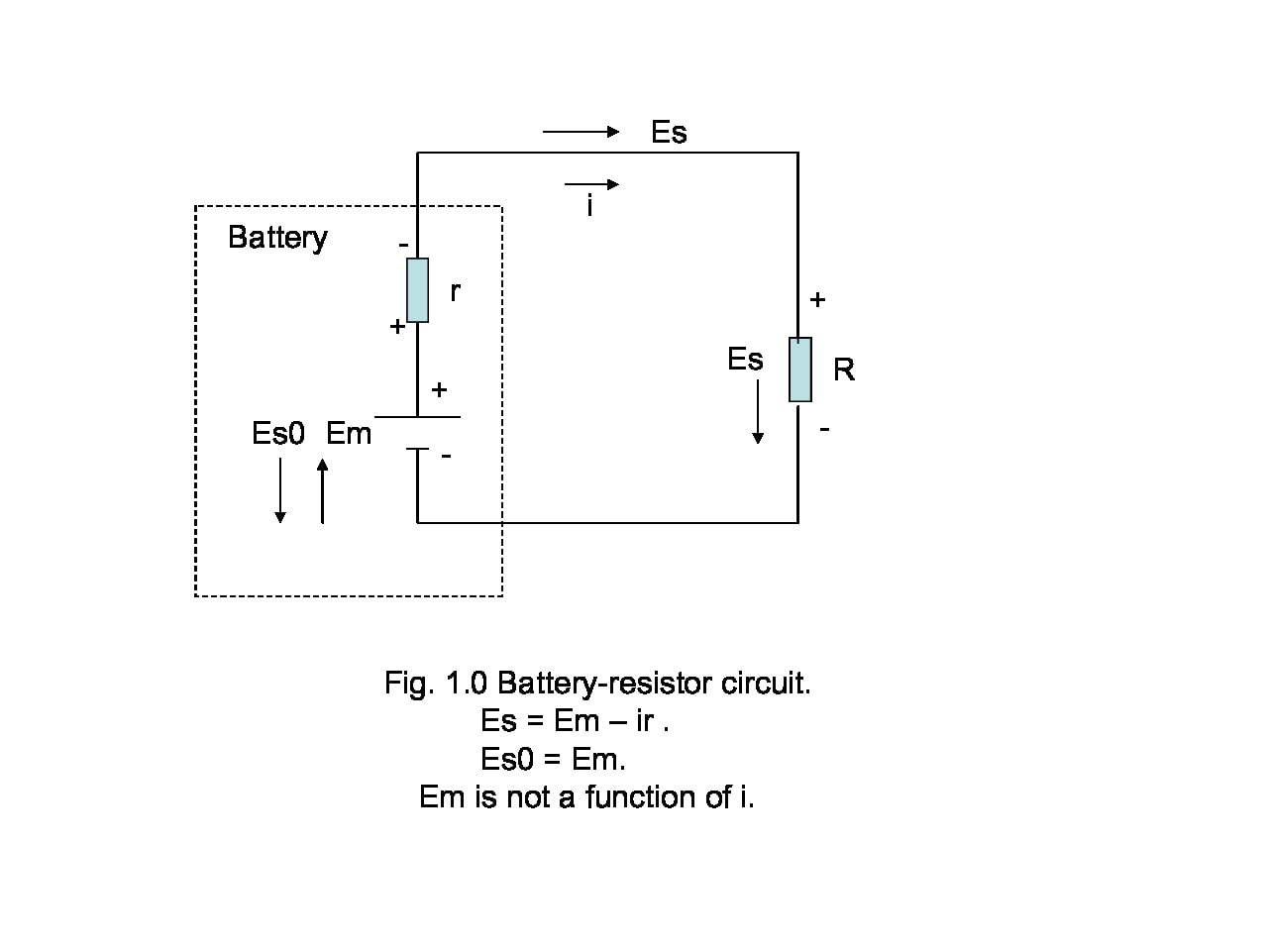
Mechanical energy conservation can be fixed if clarity replaces ambiguity within the framework of the law of energy conservation. The first step is to develop, not derive, an equation for mechanical energy conservation by specifying the two sides of the first law of thermodynamics. For the right-hand side, we write$$\Delta E_{int} = 0$$This equation implies that we have already chosen an isolated system and states that neither work nor heat crosses the system boundary. 1
The second equation provides the left-hand side,$$\Delta E_{int}= \Delta E_{kin}+\Delta U$$It establishes that having predicated an isolated system, the only appreciable internal energy changes to be considered are kinetic and potential. Combining the two equations gives the mechanical energy conservation equation,$$\Delta E_{kin}+\Delta U =0~~~~~\text{(III.1)}$$Obviously, this is not a derivation but a narrowing down of the law of energy conservation to mechanical energy conservation when internal energy transformations stay within the realm of kinetic and potential. In short, mechanical energy is conserved when it is. This may sound circular and it is, but we cannot do any better because the law of energy conservation cannot be derived from anything. Nevertheless, we can list the conditions that must be true for equation (III.1) to be applicable.
Conditions for mechanical energy conservation
Based on the development above, we can deduce that the mechanical energy of a system is conserved when all of the following conditions are met.
- The system is isolated.
- The system consists of at least two internal components.
- Any relative displacement between pairs of internal components occurs in the absence of dissipative forces, i.e. forces that convert mechanical energy into heat.
Condition 1 ensures that no external work or heat is allowed to cross the system’s boundary; if it does, the mechanical energy of the system will not be conserved. Condition 2 explicitly excludes single-component systems. Such systems, already assumed isolated by condition 1, can only maintain their state of motion because the assertion that the mechanical energy of an isolated single-component system is conserved can mean only one thing: ##\Delta E_{int}=\Delta E_{kin}=0## which is another way of expressing Newton’s first law. Condition 3 precludes mechanical energy conservation when dissipative forces mediate energy transformations and specifically excludes cases where heat is generated within the system.
Being based on the law of energy conservation, these conditions provide the needed clarity. Before entertaining any consideration of MEC, they exclude the choice of single-component systems. Furthermore, they define equation (III.1) as the starting point for mechanical energy conservation which says, in mathematical language, that “when mechanical energy is conserved, the sum of kinetic and potential energy changes within the system is zero.” The equation that we saw in part I,$$K_A+U_A=K_B+U_B~~~~~\text{(I.1a)}$$ can no longer be the starting statement of MEC, but may be arrived at by algebraic manipulation of equation (III.1). Finally, the conditions implicitly ignore non-conservative, non-dissipative forces and focus on dissipative forces that irreversibly convert mechanical energy into heat. In that regard, they allow the use of mechanical energy conservation in cases such as the roller coaster and the Atwood machine as long as the Earth is part of the system.
Work-energy theorem modification
We will now consider how to handle situations under option 2 when one or more of the conditions are not met and mechanical energy is not conserved. These could be cases of single-component systems or situations where dissipative forces significantly contribute to energy transformations or both. With option 1 such cases are handled by using the work-energy theorem. Having seen that elements of work ##dW## can be written as the scalar product ##\vec F\cdot d\vec s## and that not all such scalar products are identified as elements of thermodynamic work, we have to abandon the work-energy theorem. With option 3 the W-E theorem is superseded by the law of energy conservation. Here, we need a formulation that bypasses conservative forces. We have seen such an equation in part I, $$\Delta K =\int_A^B \vec F_{net}\cdot d\vec s~~~~~(\rm{I.2})$$It is suitable for all cases where mechanical energy is not conserved as well as to single-component systems regardless of whether dissipative forces act on them.
The general strategy would be to write equation (I.2) as the starting equation, rewrite ##\vec F_{net}## as ##\sum \vec F_i##, and evaluate the ensuing line integrals separately.2 In the case of a single-component system with no dissipative forces present, the equation may become ##\Delta K=W_{net}##, where ##W_{net}## is the sum of the works done on the system by all conservative forces plus the work done by non-conservative forces, non-dissipative forces. If such a single-component system is expanded to form an isolated multi-component system, then mechanical energy will be conserved as long as all three conditions listed above are met. In the case of a single-component system with dissipative forces present, the equation may become ##\Delta K=W_{net}+\int_A^B \vec F_{diss.}\cdot d\vec s##, where ##W_{net}## has the same meaning as above. If one deems it necessary to mention “work”, it could be “the work done against friction” or “the amount of mechanical work that is converted to heat by friction.”
Option comparison
Option 1 — Do nothing
Option 1, the “do nothing” option, preserves the status quo including its flaws and ambiguities. It is the path of least resistance because no action needs to be taken. It is likely to be selected by those who, after reading this three-part series, still think that there is nothing wrong with MEC or that, even though MEC has flaws and ambiguities, it is good enough for a “first pass”. We cannot teach everything all at once, the argument goes, therefore some allowances and compromises are acceptable – one does not need to dot the i’s and cross the t’s. After all, in most cases, there will be no “second pass”. A vast majority of physics students will terminate their physics education after the first two introductory courses. Those who do go on (about 1 in 60) to obtain a complete undergraduate physics education will revisit energy conservation in an intermediate classical mechanics course and see it done right. Therefore since the do-nothing option does not harm all around, why bother doing things differently? “If it ain’t broke, don’t fix it”.
Option 2 — Compromise
Option 2, the “compromise option”, provides fixes and moves in the right direction. It divorces MEC from Newton’s second law and views mechanical energy changes as a subset of all internal energy changes. As such, it is a first step towards bringing the law of conservation of energy to the forefront of one’s thinking. It is likely to be selected by those who are not satisfied with the status quo and cannot accept that ambiguous and confusing teaching can ever be “good enough”. Choosing option 2 requires the effort to reconfigure one’s teaching methods in terms of the more comprehensive law of energy conservation. It also requires one as part of one’s teaching by example to define the system carefully and to verify that all three conditions are met before mechanical energy conservation is invoked.
Option 3 — Replace with energy conservation
A wrinkle in the adoption of option 2 is that it requires giving up the equation (I.1a) which is used extensively in the second half of the introductory physics sequence where fields are introduced in earnest. One talks about the potential energy of a charged particle in an electric field and the potential energy of a magnetic dipole in an external magnetic field without mentioning the charges and currents that set up these fields. They are ignored in the same sense that the Earth is ignored in the “massive Earth approximation”. However, when mechanical energy is conserved under option 2, distant charges and currents must be included as part of the system in keeping with condition 2.
Textbook practice
Option 3, the “nuclear” option, repeals mechanical energy conservation as well as the work-energy theorem and replaces them with the law of conservation of energy. It is attractive because it can be widely applied to all cases involving energy transformations and it has no criteria or conditions to keep in mind. The only considerations are (a) defining the system to properly separate external work from internal energy changes and (b) enumerating the internal energy changes that matter. It is likely to be selected by those who are taken by its simplicity, its generality of scope, the range of its applications, and the realization that it can provide a coherent and comprehensive narrative across a large number of topics in introductory physics.
In textbooks, the law of energy conservation usually makes its formal appearance as the first law of Thermodynamics following the presentation of temperature, the ideal gas law, heat, and the kinetic theory of gases. At that point, mechanical energy conservation morphs into energy conservation while the work-energy theorem has already faded away. It is eminently sensible to adopt option 3 right from the start in introductory physics courses and mention heat as a change in internal energy when friction is introduced in dynamics. One can only surmise that the derivation of the work-energy theorem from Newton’s second law in textbooks is driven by a desire to set the four-lettered h-word aside by sweeping it under the rug and keeping it hidden until later. However, this radical change is not for the faint of heart as it is sufficiently removed from the mainstream to be considered perhaps blasphemous to the canon.
Final remarks
This is the point where the reader is invited to answer the question, “Which option would you choose?” I can only speak for myself. There was a time when I believed that if I could wave a magic wand and have option 3 instituted that would be a good thing to happen. I was repelled by the shortcomings of Option 1 and attracted by the simplicity and generality of Option 3. I started writing this series more than four months ago to expose the PF readers to the possibilities that Option 3 has to offer and recruit them to the Option 3 camp. However, in the intervening interval, as I thought about the usefulness of mechanical energy conservation, I began to have doubts that it should be abandoned altogether. I argued with myself that mechanical energy should be kept because it develops into the Hamiltonian in an intermediate Classical Mechanics course and the Hamiltonian becomes extremely important in Quantum Mechanics. Thus, I self-converted myself to an adherent of compromise option 2. I cannot see myself going back to the muddled thinking of the status quo. What we have now is “broken” and needs fixing.
The most important lesson that I learned from this exercise is that consideration of the law of conservation of energy is a perspective that provides clarity of thought outside the proverbial box. My recommendation to the reader is to keep in mind that the law of energy conservation is a reliable multipurpose alternative to mechanical energy conservation and the work-energy theorem.
Footnotes
1. In reality the right-hand side is approximately zero in the sense that we ignore external work or heat that gives rise to relatively small changes in internal energy. When we considered the free-falling flower pot (example II.1), the isolated system was the flower pot and the Earth. We affirmed that no work crosses the boundary, tacitly ignoring the external gravitational forces exerted on the flower pot by the Moon, the Sun, Jupiter, galaxies far far away, etc. Understanding which energy transformations are to be kept and which are to be ignored is at the core of applying the law of energy conservation.
2. Needless to say the evaluation of any line integral that involves conservative forces can be expedited by writing as the negative change in potential energy.
Discussion
I am a retired university physics professor. I have done research in biological physics, mostly studying the magnetic and electronic properties at the active sites of biomolecules and their model complexes. I have also dabbled in Physics Education research.




Buying medicine online is very easy.
It’s possible to browse at any time without stepping out of home.
Internet pharmacies allow you to easily compare prices and find the best deal.
vidalista forum
Your order is shipped straight to your door, saving time and effort.
Moreover, there is no requirement to stand in long lines at a brick-and-mortar store.
For those with ongoing illnesses, auto reorders ensure therapy stress‑free.
In short, online ordering for meds saves your hours and peace of mind.
В наше время кроссовки обрели невероятную известность везде.
Такую обувь выбирают люди всевозможных возрастов и родов занятий.
Основная причина столь всеобщей любви — комфорт и практичность.
https://fashionessa.ru/
Известные марки регулярно представляют свежие коллекции, усиливая интерес.
Кроссовки надёжно закрепились в уличную культуру и повседневность.
Год от года сезоном их популярность лишь возрастает.
Ставки на спорт стали огромную привлекательность в наше время.
Ключевой фактор этого заключается в варианте переживать незабываемый эмоциональный подъем.
Во время просмотра матчей ставки приносят невероятные ощущения к игре.
Поклонники спорта способны переживать за любимых игроков, а также получать прибыль на результатах.
Современные букмекерские сайты обеспечивают процесс ставок простым плюс быстрым для широкой аудитории.
Множество вариантов спортивных событий позволяет каждому болельщику выбрать то, что нравится.
Таким образом букмекерские ставки делают спорт просмотр матчей гораздо более захватывающим а также волнующим.
https://rick.ulmoda.ru/futbolnye-stadiony-arkhitektura-i-atmosfera-3923458/
”
Every physics class begins by saying “what we taught you last year was wrong”.
”
Feynman’s lament. He hated it, but try as he might could see no way out of it.
Thanks
Bill
I would replace it with option 3 – but use option 1 and 2 as examples. Then I would pull the rabbit out of the hat and show it is a result of Noethers Theorem. I think Morin does something similar in his textbook:
[URL]https://scholar.harvard.edu/david-morin/classical-mechanics[/URL]
I really like Morin – the problems are very hard – but he has many of the solutions in the book. The difficulty of the problems does to some extent overshadow its pedology which I like a lot. It’s even approachable by an upper level HS student after they have done calculus – Morin even thinks they would have a hoot – and so do I. But the teacher should guide them through the difficulty of the problems – they definitely are well above what is expected of a HS student. Excellent preparation as well for my all time favourite mechanics textbook – Landau – Mechanics. I would love if that could be taught HS – but unfortuneately that would be expecting far too much. First year college after Morin – maybe – but HS – no.
Thanks
Bill
I have not used Thornton et al. although I am sure I considered it for adoption at some point. Attempts have been made to depart from the canon, but as far as I know, that’s what they remain, attempts.
”
Thanks for a very thoughtful series.
I am absolutely in the option 3 camp. The reasons I see for the other options are largely pedagogical in nature, and in my experience (both as student and professor) serve to confuse more than to enlighten. An endless semantic jungle.
”
I agree with all that. But tell me, as a professor, are you prepared to present option 3 in the classroom? I avoided doing that when I had the chance because because I deemed that it would be easier to reach my students if I went along with the textbook than if I said “here is a better way of handling this, don’t look at the textbook,” Most beginner students dislike that and become less receptive when it is done to them, especially those who have paid good money for a textbook. Besides, I don’t believe there are (m)any textbooks that have embraced option 3.
The use of the Atwood machine as a counterexample in the first article is flawed. It confuses “if” with “if and only if”. As Wikipedia says, we can derive the correct use of energy conservation for the Atwood machine by considering the Lagrangian in the presence of constraints.
[URL]https://en.wikipedia.org/wiki/Atwood_machine[/URL]
[URL]http://farside.ph.utexas.edu/teaching/336k/Newtonhtml/node79.html[/URL]
I would keep option 1, except that the ambiguity is not a flaw, but a feature.
The work-energy theorem of classical mechanics does not refer to the same work as in thermodynamics. The great thing about the ambiguities are that it highlights that subjectivity is required in thermodynamics, because thermodynamics is not fundamental, but comes from a coarse graining, which depends on what one is measuring with what accuracy. This idea of coarse graining lays the groundwork for the renormalization group and effective field theory. Also, the subjectivity prepares one for the Copenhagen interpretation, where subjectivity is needed to determine what an observer or measurement is. These lead to questions as to how thermodynamics is related to some presumably more fundamental theories like mechanics. And analogously as to whether quantum mechanics might point towards the existence of a more fundamental theory. :oldbiggrin:
This is a good conclusion to your series!