Frequently Made Errors in Climate Science – The Greenhouse Effect
Table of Contents
1.What is meant by “The Greenhouse Effect”?
Many gases, such as H2O, CO2, CH4, are transparent to visible light but absorb and emit parts of the infrared spectrum. Most of the visible light reaching the Earth’s surface gets re-emitted, eventually, as infrared. Media that pass visible light through but block infrared can act as heat traps.
2. Real Greenhouses
X “The Greenhouse Effect does not exist; Prof R.W.Wood proved it in 1909.”
Most glass also blocks parts of the infrared band. It was widely believed that this was primarily responsible for the effectiveness of greenhouses.
Prof. Wood suspected that blocking convection was the primary mechanism, so set up a simple experiment to test this. There are a number of weaknesses in the experiment, but the essential conclusion is correct: real greenhouses work primarily by blocking convection.
✓ “Whether or not the Greenhouse Effect exists, it is not the main way real greenhouses work”
However, this just means that the term ‘greenhouse effect’ may be misleading. Wood’s result says nothing about how the atmosphere works.
3. Black Body Earth
If we treat the Earth as emitting and absorbing radiation as a “black body”, ignoring the atmosphere, and treating incoming light as spread evenly over the whole Earth’s surface all the time, we can calculate the equilibrium temperature as -18C. At that temperature, black body radiation would balance insolation.
Adding a non-greenhouse atmosphere, e.g. pure nitrogen doesn’t change this. The atmosphere would take no part in the energy balance. By conduction, it would come to match the surface temperature of the Earth, throughout its depth.
Note:
- With a non-greenhouse atmosphere but now allowing the realities of a rotating sphere, convection would boost the upper atmosphere to something approaching the temperature of the hottest spot on the surface. The surface layer of the atmosphere would be a little cooler by virtue of conduction back to the cooler surface regions.)
- The non-greenhouse atmosphere may also result in some attenuation through Rayleigh scattering. The nitrogen in Earth’s atmosphere may scatter about 4% of light power back into space, taking the temperature down by maybe 3K.
4. The Troposphere
The atmosphere has many layers, featuring quite different processes and temperature profiles.
The atmospheric convection with which we’re familiar only operates up to the tropopause, the top of the troposphere – the band where weather happens. Beyond that, temperature inversions inhibit convection.
X “The ‘greenhouse gases’ are a net coolant since convection carries the heat through the troposphere, past 80% of them. They then block reradiation back to the surface.”
It’s the 20% above the tropopause that matters. This makes the tropopause warmer and/or higher. Since convection is limited by the lapse rate, a higher or warmer tropopause leads to a correspondingly warmer surface.
The existence of this “tropospheric hotspot” is considered a fingerprint of Global Warming.
5. Temperature and Pressure
X “It’s hotter at lower altitudes because it’s at a higher pressure, and compressing a gas heats it”
Compressing a gas heats it, but won’t keep it hot. If the atmosphere were just a static layer of gases, only heated or cooled by conduction, it would all come to the same temperature.
6. The Lapse Rate
X “It’s hotter at lower altitudes because if air rises it expands and cools”
This only explains why convection cannot bring the troposphere to a uniform temperature. It does not explain why there should be a temperature difference in the first place.
Since conduction is not limited by the pressure gradient, there must be an active process producing the temperature gradient. This process is the heating of the Earth’s surface by the sun.
The full story of energy transfers is quite complex. See Trenberth and Kiehl, 1997, Fig 7. Omitting all the absorptions and reradiations:
- The Sun warms the Earth’s surface
- The heat energy is transferred back to the air in the troposphere by a mix of conduction, convection and radiation. Overall, 60% makes it up through the troposphere at least partly by convection, 40% by radiation only.
- Convection’s ability to carry up the heat is limited because of the pressure gradient: rising air expands and cools. The resulting temperature gradient is known as the Lapse Rate
- The troposphere is the layer in which convection can operate. At the top (the tropopause), the temperature gradient is insufficient.
X “We can calculate the surface temperature from the height of the tropopause, the temperature there and the lapse rate. This fixes the surface temperature.”
That has causality backward.
✓ If the mean surface temperature changes, the height of the tropopause will change.
7. Greenhouse Gases
X “There’s nothing special about CO2. All gases can absorb heat”
All gases can conduct heat, but the ability of a molecular species to absorb and emit radiation depends on the intervals in its internal energy states and the polarity of its structure.
If vibration of the atoms in a molecule does not involve a net oscillation of electric field then that vibration cannot absorb or emit electromagnetic radiation. Diatomic gases like N2 and O2 are non-polar (or”homo-polar”). A vibration in the bond between the atoms does not result in any net movement of charge. Other energy levels of those molecules do not have the right intervals to interact with light in either the visible or infrared bands, so are completely transparent to both. Only much higher energies, sufficient to ionize the gases, would be strongly absorbed.
Both water and CO2 are hetero-polar, so can act as dipoles. Some vibrations involve a negatively charged atom group moving one way while a positively charged group moves the other. This net oscillation of charge allows them to interact with radiation at certain frequencies.
X “CO2 is insignificant compared with H2O as a greenhouse gas”
H2O, CO2, CH4 and many others can absorb/emit in parts of the infrared. None of them do so in the entire infrared band. Increasing the level of relatively rare greenhouse gas has more effect than increasing the level of a more common species.
8. Forcings and feedbacks
Climate scientists distinguish factors affecting global temperature as either forcings or feedbacks.
X “Atmospheric H2O is a forcing that overwhelms CO2”
A feedback is a variable which both affects temperature and is affected by temperature. Pure feedback would be a variable entirely controlled by temperature.
These can be further divided into negative and positive feedbacks. This list is by no means exhaustive.
- A few positive feedbacks
- Atmospheric H2O: the warmer the atmosphere, the more water vapor it will hold.
- Polar albedo: as ice caps melt, less incoming light is reflected straight back through the atmosphere.
- The warming of soils and oceans can lead to the release of CO2.
- A negative feedback
- The hotter the Earth’s surface, the more infrared it emits
Note: If something is a negative feedback it acts to dampen change; it cannot make the change go in reverse. That said, delayed feedback can lead to cyclic and chaotic behaviours.
A pure forcing is a variable which affects temperature but is not affected by it. Radiative output from the Sun clearly fits that description.
More loosely, a variable tends to be called a feedback if it is primarily controlled by temperature, and forcing if primarily controlled by other factors. On that basis, anthropogenic CO2 is forcing, but H2O is a feedback.
✓ Anthropogenic CO2 is a significant force because its effect is amplified by positive feedbacks, such as water vapor.
9. The Greenhouse Effect is Logarithmic, roughly
“The light passing through a filter should fall as the negative exponential of the optical thickness, so why is the effect logarithmic?”
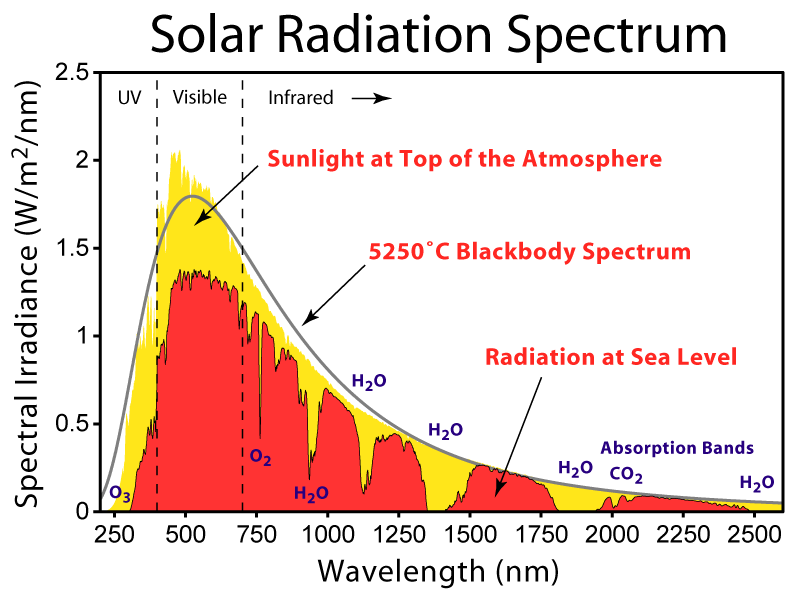
“Solar Spectrum”. Licensed under CC BY-SA 3.0 via Wikimedia Commons – http://commons.wikimedia.org/wiki/File:Solar_Spectrum.png#/media/File:Solar_Spectrum.png; but note the 5250C is incorrect, it is closer to 5777K.
The diagram shows the absorption bands (yellow) for incoming radiation, and the same applies to outgoing. It shows that for most of the width of an H2O or CO2 band, absorption is already substantial.
The quantum basis for specific bands suggests, at first sight, that the wavelengths should be quite precise. However, some subtler processes spread the bands. In particular, the Doppler effect means that molecules moving towards the radiation absorb at a shorter wavelength, while those moving away absorb at a longer.
The primary consequence of adding more of an already abundant gas is to increase the number of molecules at the most extreme speeds relative to the radiation. This increases absorption at the edges of the band, broadening the band slightly. It is this that grows logarithmically with the prevalence of the gas.
“How can it be logarithmic? That would mean adding the first little bit of a new gas would have an infinite effect.”
The logarithmic relationship would not apply for a rarer gas that is still well short of full absorption in the center of its bands. Likewise, it breaks down when bands broaden so much that they overlap.
Masters in Mathematics. Interests: climate change & renewable energy; travel; cycling, bushwalking; mathematical puzzles and paradoxes, Azed crosswords, bridge



[QUOTE="mheslep, post: 5518501, member: 70823"]Yes CO2 molecules radiate per Stephan-Boltzman law based on their own temperature like everything else, but the scattering of IR is a different phenomenon and is independent of the temperature of the gas. Think IR "mirror", though with an arbitrary angle of reflection. Yes mirrors have their own blackbody temperature as any IR thermometer will confirm, but this has nothing to do with the ongoing reflection of light, and changing the temperature of the mirror won't change its reflective properties.http://scied.ucar.edu/carbon-dioxide-absorbs-and-re-emits-infrared-radiation“And again we get problems from the use of metaphorical comparisons. Reflection is very different to absorbed energy that is re-emittid to the whole surrounding as a result of internal intensity.The co2 shows absorbing by decreasing emitted spectral intensity, that means lower temperature. It absorbs the most intense spectral fraction at a spectral intensity in emitted radiation of about 220K. It absorbs high intensity and emits it with low intensity. That means that it needs more energy to emit the same intensity as the surface emits.
[QUOTE="Reality Is Fake, post: 5519749, member: 598755"]It shows 161W absorbed from insolation. We can measure suns irradiative intensity to 1000W in large parts.”Does your solar panel collect 1000W when you lay it flat on the Antarctic or during the night? The infographic shows global energy balance, not energy incident on a solar panel angled perpendicular to solar rays at noon!
[QUOTE="haruspex, post: 5518634, member: 334404"]I don't know where you got 161 from.”It shows 161W absorbed from insolation. We can measure suns irradiative intensity to 1000W in large parts. I think solar cells are calibrated to around that amount. And they don`t absorb longwave radiation as a source of energy.”The diagram Bandersnatch posted has 343 W/m[SUP]2[/SUP].”Not at the surface. And why use 343W which would represent a long timespan average when you calculate an intensity in a state. 343W is the effect calculated from a temperature and temperature is measured intensity. Effect/m^2 as a value of radiating intensity is a dimensionless expression describing energycontent in matter.It is fundamentally wrong to use an average derived from energy received over time per total area, when analyzing the earths exchange of energy with the sun when it´s heated only at half the area that it uses to transfer heat, and temperature is something that describes intensity independent of time.Temperature of earth surface is a state that is a product of the immediate relations to the surroundings, it is totally dependant on the constant feed of energy from the sun at any point in time. Temperature of the surface does never increase without suns radiation. The only thing raising temperature is insolation. Not matter.”The flux density from the Sun at Earth's radius is 1370. This is captured by the Earth over a radius R[SUB]E[/SUB], i.e. over a disc, so area πR[SUB]E[/SUB][SUP]2[/SUP]. If we pretend this is uniformly distributed over Earth's surface, 4πR[SUB]E[/SUB][SUP]2[/SUP], that's 343W/m[SUP]2[/SUP].Allowing for non-uniform distribution puts the average temperature down a bit.”And without an atmosphere 1370W would irradiate every m^2 of irradiated surface area. Now, with an atmosphere we can measure around 1000W/m^2.When calculating absorbed intensity of radiation at the surface, the intensity that is the cause of surface temperature in a heat transfer and is directly proportional to intensity emitted, what use do we have of an average?Why would we calculate emitted intensity which is equal to surface temperature, with surface area that is not heated and only is cooling?We have measured values of absorbed intensity thats fits emitted intensity, and we have measurements showing how low intensity the atmosphere reaches. We find the balance from absorbed intensity/m^2 and that is transformed into an emitted intensity in double the surface area. The atmosphere is irradiated at same surface area that emitted from earth surface, It reaches an intensity lower than the surface as it is low in density and distributes all absorbed energy in all its volume from any surface point. Average of emitted intensity is a correct value of average cooling of 1m^2 from absorbed intensity from 2m^2.That puts the balance over heavily to cooling ability in relation to heat absorption ability.The number 343W belongs in an energybudget that describes energy use over time in the system, and should be accompanied by a unit of time, while radiative balance which is what defines temperature, uses W/m^2 or simply W for realtime state.Using 343W in radiative balance calculation or energy balance will give a flawed model for heating and cooling, implying that there is a sensitive balance easily moved. Earth is cooled at double the efficiency it is heated, as all absorbed heat of 1m^2 is emitted from 2m^2. The cooling mass of solid planet is double the heated mass of solid planet.Why use a value of not heated mass to calculate the radiative cooling resulting from heated mass?The heat transfer that we observe happens at a rate of about 1000W, not 343W, and the cooling at around 350W as a result of poor emissivity of the surface, and the atmosphere emissivity is causing more loss in intensity radiating at an intensity of about 220W in average below the tropopause. To raise surface intensity it must have higher intensity, above 350W/m^2, emitted in both directions from 2 m^2, as it emits only half of absorbed energy towards the surface.According to the colorful arrows and very unrealistic numbers displayed as "energy budget" using energy absorbed as a function of time as the value of realtime intensity in units/m^2, we can see that an absorbed amount of 343 at the TOA is transformed into total ~430W in the atmosphere that radiates a much larger part to earth than space. There is no way to increase the amount of energy in the system from 343 to 430 total just by an absorbing gasvolume between the source and the surface. The atmosphere is absorbing an amount of 430W/m^2 from 343W/m^2 and radiates 430W/2m^2=860W/m^2 since it is claimed to radiate in two directions”
[QUOTE="Reality Is Fake, post: 5519676, member: 598755"]sun is the only relevant source of radiation in the area.”That is merely your opinion. It is unsupported by the facts. Do the math. A black body surface at -18C emits 343 W/m[SUP]2[/SUP], as much as the average reaching Earth's surface from the Sun. If 'empty' space were at 130K say, instead of 4K, it would add 20W/m[SUP]2[/SUP] (I think that's about right), which would be a significant addition.[QUOTE="Reality Is Fake, post: 5519676, member: 598755"]maybe it is absorbed.”Of course it must be.[QUOTE="Reality Is Fake, post: 5519676, member: 598755"]it is not having any measurable effect on intensity in the hot body.”That is merely your opinion. It is unsupported by the facts.
[QUOTE="haruspex, post: 5518491, member: 334404"]By what magic would the Earth's surface absorb emissions from the sun but from no other emitter?”By the magic that sun is the only relevant source of radiation in the area.No other radiating body in the surroundings or in contact with the planet surface has an intensity that can have an effect that raise earth´s radiating intensity.”Simple thought experiment: two black bodies in close proximity at different temperatures. The cooler body emits radiation as per S-B law. Some of this is in the direction of the hotter body. What, in your view, happens to that radiation?”I´m not sure, and as far as I know noone is. The common description is that radiation at the scale of photons reach the hotter surface as a photon is not restricted to net flux direction, and maybe it is absorbed. What we can measure in the states of the two bodies is very clear and consistent relative development, that tells us that no matter what happens to photons emitted in direction towards the hot body from the cool body, it is not having any measurable effect on intensity in the hot body.We can confirm by observation that cold photons absorbed in a hot surface is an unmeasurable totally insignificant theoretical process, and we must not include it when we calculate heat transfer at all. We know for sure that it does not raise temperature, all observations in real processes of all kinds of heat transfer shows that cold photons has a nonexistant effect on a warm body.
[QUOTE="rbelli1, post: 5518119, member: 315621"]The ammonia is irrelevant to the fact of cooling. The system would allow cooling as a simple block of metal. The ammonia just speeds the transfer of heat.”That is precisely my argument concerning the G-H hypothesis, The atmosphere speeds Heat transfer up from surface to space, by adding a radiating volume and transfer of heated mass from irradiated parts to the cooling parts, and blocks it from sun to surface. That means that it regulates the amount of irradiated energy by cutting off the most high frequent wavelenghts and absorb parts in the spectrum at several other wavelenghts. On top of that, it reflects others like white in clouds.Some good chunks in solar spectrum are heating the volume, atmosphere, and lets earth radiate at cooler temps as well as shielding it. That makes the atmosphere a limiting effect on the surface temp from insolation. A limiting effect on thermic radiation.Is it reasonable that a dampening structure in a heat exchange is increasing intensity in the heated body?Is it probable that a property of the surface of a radiating body that decrease intensity in absorbed high thermic radiation, has an opposite effect in low intensity thermic radiation?Can an ice cold gasskin that gets all it´s heat from the sun, mainly absorbed and emitted insolation from the surface, perform work on same surface by receiving energy in heat transfer and reaching a much lower intensity state. The atmosphere loses energy that is absorbed from OLR at any measured point above the surface boundary. Temperature=intensity, is massively decreasing with distance as radiation interacts with it. That is very strong dampening of radiation, and that correlates with cooling – low effiancy through lossy properties like lacking heat capacity. The atmosphere is really bad at absorbing thermic radiation in relation to the surface.When a radiating body is suspended in a vacuum of 3K without boundary preventing radiating to space, will rise in intensity when it has a surface layer of ice cold fluid lowering the radiating total intensity?The total absorbed energy is distributed in a larger mass at lower total intensity in a planet with atmosphere, than in a planet with solid surface and no fluid cool surface layer.Something that is characterized by distributing a fixed amount of energy in a bigger volume at lower concentration, both in received and radiated energy, is not a property that increase intensity. It results in lower intensity at any point measured from surface to space in emitted longwave and from space to surface in irradiated shortwave.”The second statement says cooling = 0 if only radiative cooling. The first says radiative cooling is possible. Please at least be consistent is your technobabble.BoB”If convection is added by absorbed insolation and/or OLR and results in a bigger volume mass of lower total intensity = lower total temperature.The atmosphere absorbs a fraction of earths total emitted radiation and reaches a much lower intensity, The difference to a non existant atmosphere is more mass at lower density radiating at lower intensity.My point was that an atmosphere is only cooling if it adds mechanisms for heat transfer to space that lowers concentration of energy in the total mass. And I think that is the only effect an atmosphere can have on received energy from an external source.Actually, the only cooling possible is radiative cooling, convection and other processes is matter absorbing energy in the radiation field and gets shaped by it.
[QUOTE="D H, post: 5518646, member: 42688"]You placed the "GHG lives here" sticky at the wrong point in the picture, which might be why you are confused. The greenhouse gases "live" over on the right, and are already labeled greenhouse gases.”You didn't read my postI was showing that latent heat provides a shortcut back out of the atmosphere bypassing greenhouse gasses for about 1% of total heat that made it to earth's surfacebecause from reading that report i took away that they've modelled heat fluxes as either/or not morphing over here on the right ghg should be down low where i drew it because unlike surface radiation, latent only has to traverse ~20% of the ghg which was the point of my pressure vs altitude chart
[QUOTE="jim hardy, post: 5517968, member: 327872"] “You placed the "GHG lives here" sticky at the wrong point in the picture, which might be why you are confused. The greenhouse gases "live" over on the right, and are already labeled greenhouse gases.
“You placed the "GHG lives here" sticky at the wrong point in the picture, which might be why you are confused. The greenhouse gases "live" over on the right, and are already labeled greenhouse gases.
[QUOTE="Reality Is Fake, post: 5518046, member: 598755"]Well, since the surface temp would be heated with almost 400K without an atmosphere, it´s definately cooling incoming.”That is incorrect. Without an atmosphere, the Earth's surface temperature would be below freezing. The incoming solar radiation is the solar constant, 1365.2 W/m[sup]2[/sup], divided by 4, or 341.3 W/m[sup]2[/sup]. You apparently forgot to divide by 4. The reason you need to divide by 4 is because the Earth's cross section to incoming solar radiation is that of a circle with a radius equal to that of the Earth, or [itex]pi R^2[/itex]. The Earth's surface area is that of a sphere with a radius equal to that of the Earth, or [itex]4pi R^2[/itex].Some of that incoming radiation will be reflected away. Assuming an albedo of 12.5% (the average albedo of land and ocean), only 298.6 W/m[sup]2[/sup] will go into heating the Earth. That yields a blackbody temperature of 269.3 kelvins. Assuming an albedo of 30% (the albedo with clouds), the blackbody temperature drops to 254.7 kelvins. The world would be an inhospitable place were it not for the greenhouse effect.”I have seen that too many times, it stings my eyes.”Instead of complaining, you should try to understand that diagram. It is fundamentally correct.”If the atmosphere would give 333W it would have to have an average temperature of 4√(333/0.0000000567)=276KWe can be very sure that it doesn`t.”We can be pretty sure it does.You have been making a number of false claims. Please read the rules that pertain to climate change.
[QUOTE="Reality Is Fake, post: 5518046, member: 598755"]The number of 161 hitting the earth is ridicolous,”I don't know where you got 161 from. The diagram Bandersnatch posted has 343 W/m[SUP]2[/SUP].The flux density from the Sun at Earth's radius is 1370. This is captured by the Earth over a radius R[SUB]E[/SUB], i.e. over a disc, so area πR[SUB]E[/SUB][SUP]2[/SUP]. If we pretend this is uniformly distributed over Earth's surface, 4πR[SUB]E[/SUB][SUP]2[/SUP], that's 343W/m[SUP]2[/SUP].Allowing for non-uniform distribution puts the average temperature down a bit.
[QUOTE="Reality Is Fake, post: 5518046, member: 598755"]The number of 161 hitting the earth is ridicolous, we can measure it to 1000. Why use such a misleading number, of course you will get a conclusion that is way off.”-There is a night side-Earth is not a circle but a sphere
[QUOTE="jim hardy, post: 5517968, member: 327872"]Much of that 80 watts of latent heat gets deposited above 80% of the greenhouse gas [ATTACH=full]103078[/ATTACH]where its transport mechanism changes from convection to radiation, both upward and downward of course,[ATTACH=full]103079[/ATTACH]i've not been able to figure whether they model it that wayDownward bound has to get back through the ghg layer., upward doesn'tfrom last pageold jim”If I understand your reasoning, increased GHGs should just lead to a hotter tropopause. But as we know, the lapse rate represents the limit on the ability of convection to even out the temperature. So a hotter tropopause at the same altitude, or the same temperature tropopause at a greater altitude, means correspondingly hotter at ground level.The uncertainties over clouds' affect on downward radiation do not invalidate the principle of the greenhouse effect, they merely make its strength hard to assess. Until such time as clouds are better understood, we must a) look at direct measurement and b) apply risk analysis.Direct measurement from satellites shows a significant excess of incoming radiation – especially considering that for a stable temperature the net flow should be outwards.
[QUOTE="Reality Is Fake, post: 5518046, member: 598755"]I have seen that too many times, it stings my eyes. If the atmosphere would give 333W it would have to have an average temperature of 4√(333/0.0000000567)=276KWe can be very sure that it doesn`t.”Yes CO2 molecules radiate per Stephan-Boltzman law based on their own temperature like everything else, but the scattering of IR is a different phenomenon and is independent of the temperature of the gas. Think IR "mirror", though with an arbitrary angle of reflection. Yes mirrors have their own blackbody temperature as any IR thermometer will confirm, but this has nothing to do with the ongoing reflection of light, and changing the temperature of the mirror won't change its reflective properties. http://scied.ucar.edu/carbon-dioxide-absorbs-and-re-emits-infrared-radiation
[QUOTE="Reality Is Fake, post: 5518010, member: 598755"]If emission is constant, nothing more is absorbed than what it gets from the sun.”By what magic would the Earth's surface absorb emissions from the sun but from no other emitter?Simple thought experiment: two black bodies in close proximity at different temperatures. The cooler body emits radiation as per S-B law. Some of this is in the direction of the hotter body. What, in your view, happens to that radiation?
[QUOTE="Reality Is Fake, post: 5517989, member: 598755"]I think I saw liquid ammonia in there somewhere;)”The ammonia is irrelevant to the fact of cooling. The system would allow cooling as a simple block of metal. The ammonia just speeds the transfer of heat.[QUOTE="Reality Is Fake, post: 5517989, member: 598755"]Radiative cooling is very possible,”[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]It is only cooled when another way of transfer is added to radiation”The second statement says cooling = 0 if only radiative cooling. The first says radiative cooling is possible. Please at least be consistent is your technobabble.BoB
[QUOTE="Bandersnatch, post: 5517929, member: 399360"]Sorry for the late response to my part of the conversation. I didn't want to make this a wall of quotes (and kinda hoped haruspex would take over my bit as well), but I couldn't figure out any other way to go about it. I'll try to summarise the crux of the issue as what I think it is first, and then address some of the particular misconceptions in the spoiler.”No worries, I´m just very happy to discuss this with people that have a good manner and knowledge about the principles at work.”The last sentence here seems to be the culprit here. You're seeing the atmosphere as an additional heat sink – i.e., the idea seems to be that since for a set amount of incoming radiation Z, surface radiates X energy to space, then if we add a medium that will remove extra Y energy from the surface, and carry it away where it will then escape into space, it should mean that we've added another sink, so that the energy escaping is X+Y, meaning the radiative emissions have to go down, meaning the equilibrium temperature at the surface must become lower.”Well, since the surface temp would be heated with almost 400K without an atmosphere, it´s definately cooling incoming. And it´s not as much an extra heat sink for emission, as the surface emits only according to it´s own temperature. But it it is still cooling the surface even if the surface cools anyway. The fact is that earth cools from a big volume with an atmosphere instead of a surface only. And it would be much warmer without an atmosphere. The problem is that we treat is as two bodies when the atmosphere is actually a porous low density surface that lowers the surface absorbing ability, the emissivity-epsilon.”The issues with this picture are:- the atmosphere is for the most part not transparent to the outgoing radiation, so it can't just escape into space – it gets absorbed and reradiated in all directions, including downwards. The actual atmospheric window for escaping radiation is just about 40 W/m^2.- the thermal heat transfer (conduction, convection, evaporation) from the surface is 1. small when compared with radiative transfer, and 2. ends up being reradiated in upper parts of the atmosphere, again including back to the surface.”I have a problem with the description of bad emissive properties and high temperatures, as high temperature is an effect of good emissive properties, according to Kirchoff. "What is absorbed is emitted" he concluded, and I am not willing to abandon such a well known law unless very, very good evidence. And I think we all know that evidence of the GH-theory and co2:s heating in the atmosphere is pretty much non-existant. Even though it should be very easy to prove if a gas in an open system could do that type of work.”This could still be a valid objection if the energy balance at the surface was a net removal of energy, and would require quantifying – if it hasn't been done many times already. E.g. see the following paper (with its inforgraphic reproduced below):http://journals.ametsoc.org/doi/pdf/10.1175/2008BAMS2634.1 All that energy that was removed from the surface and then returned in the form of back radiation changes the energy balance at the surface so that there is more incoming energy Z, and the equilibrium has to change to increase radiation (or thermals, but their magnitude is secondary), meaning increase in temperature of the surface.”I have seen that too many times, it stings my eyes. If the atmosphere would give 333W it would have to have an average temperature of 4√(333/0.0000000567)=276KWe can be very sure that it doesn`t.And since surface T is Tsun=εδT^4 and atmosphere is T-surface=εδT^4. The atmosphere would have to heat the sun if it heats the earth surface because it always is a one way process.The number of 161 hitting the earth is ridicolous, we can measure it to 1000. Why use such a misleading number, of course you will get a conclusion that is way off.I have still not seen a calculation including the whole chain that can explain how a cold atmosphere can heat a warm surface. Back radiation is about photons from single molecules and that is not possible to calculate at this scale. We should stick to what we know works, heat transfer and thermic radiation. That tells us that a body radiaties only according to its own temperature.”So, looking at the atmosphere as a heat sink is faulty reasoning – it is an insulator.”Insulators is a specific thing, it does not have steep gradients, it has very low gradients.”But all you really needed, in order to know that the atmosphere is raising the surface temperature, is to do the calculations for the blackbody equilibrium temperature for an airless barren planet at 1 AU around the Sun. Since that is lower than what we've got here, it is a clear indication that the atmosphere is responsible for raising the temp. All that remains is to figure out how (which the paper linked above does nicely).”I think they calculate with average watts, not what is hitting earth in reality, 1370W. That is almost 400K. Look at the moon, above 100C.”The Stefan-Boltzman law concerns only radiative energy transfer from a black body. You can't use it for thermals.”It´s the base for heat transfer, there is no process that can change it in an open system. And earth surface is cooling to space with a ΔT to 3K, the stuff in between is details that can´t change that without a boundary with conduction.And the whole base for greenhouse theory is about radiation, so where does that leave us?And please tell us what to use then. A chart with numbers of Watt is a mockery of thermodynamics without calculations. And the numbers is clearly wrong, the atmosphere is not that warm. Even though you claim S-B is not correct for the transfer, it IS correct for the temperature.”No! The violation would be if there was colder atmosphere heating up hotter surface, whereas what we've got is the hot Sun heating up the surface.When considering the Earth+atmosphere system, you don't get any NET heat flow inward. Heat is always flowing away from the hot source (surface) to colder surroundings (including space). But there is extra energy coming inward from the atmosphere that wouldn't be there without air, which means that the NET heat flow is lower and the equilibrium temperature at the surface has to self-adjust to re-emit that extra energy.”But heat transfer is a process where the energy in the colder body does not influence the hot body in any way. The hot body radiates only according to it´s own temperature, and as you said, that comes from the sun. There is not a single observation in nature or in experiments where we can see such a process.”You should forget everything you wrote about GR there and there on after, since it's completely misappropriated, and just wrong.Forget about the c^2 in the equation, it's confusing you. It's just a unit conversion factor, and you can freely choose units in which it's equal to 1, so that all the equation says is that mass of a body at rest has some associated energy.”Now you just make me more interested, that was a sore toe.”You're mostly talking about energy conservation anyway, which is not violated when putting CO2 into the atmosphere (because the mass was already there, only not in the atmosphere) nor when increasing temperature, because the system is not closed – i.e. the Sun provides energy. If you would design a rather implausibly good insulation system for the planet, you could raise its temperature as high as the temperature of the Sun's surface, and it wouldn't violate any conservation nor thermodynamic laws.”Absolutely not. Give me the calculations. You are ignoring the distance. Please, up the level.”YWhy would CO2 on Earth heat the Sun? It doesn't make any sense..”Because it is a chain of δT^4 that goes only in one direction, and you are putting in energy at the end exhaust.”No, that is a tell-tale sign of a good insulator.This might be another significant issue – what you seem to consider 'cooling' is just heat transfer from hot to cold. Remember that we're talking about systems in thermal equilibrium with their surroundings, which include heat sinks and heat sources. In such a system there will always be temperature gradients, but this doesn't necessarily mean there is cooling”Well then, show me a good insulator in an example that has a steep gradient from a hot surface.”E.g. if you put on a sweater in winter, a steep gradient from your skin to the air will set up. By your usage of the term, the sweater is 'cooling' your skin, because you're loosing heat through it.”You have to buy new sweaters. The sweater is not hot if it keeps you warm.”For something to be cooling a body heated by an external source, it has to increase energy transfer from the body, so that more net heat is removed than without it, and the equilibrium temperature drops as a result.E.g., if you put a radiator on your processor, it'll remove more energy by increasing surface area in contact with air, reducing its temperature – i.e. a cooling effect. Conversely, if you glue a block of fibreglass to the processor, it'll slow down heat transfer, heating it up. The second case will have a steeper temp gradient than the first, but it will be undoubtedly heating.”So, youre saying that the atmosphere is like fiberglass?It really is a big problem with the comparisons using sweaters, blankets and fiberglass. It shows you have a hard time yourself to validate the theory.Can you show me an open system without fiberglass that gets warmer by being in contact with a much cooler gas… that is in contact with an optimal heat sink?The claim has to be confirmed by an observation in natural setting in scale. Why is it not possible to demonstrate in experiments?There is no proof of the concept. You say that a hot body surrounded by 3K is insulated by a lowdensity, low heat capacity gas. Why cant we design that and make it work then?
All that energy that was removed from the surface and then returned in the form of back radiation changes the energy balance at the surface so that there is more incoming energy Z, and the equilibrium has to change to increase radiation (or thermals, but their magnitude is secondary), meaning increase in temperature of the surface.”I have seen that too many times, it stings my eyes. If the atmosphere would give 333W it would have to have an average temperature of 4√(333/0.0000000567)=276KWe can be very sure that it doesn`t.And since surface T is Tsun=εδT^4 and atmosphere is T-surface=εδT^4. The atmosphere would have to heat the sun if it heats the earth surface because it always is a one way process.The number of 161 hitting the earth is ridicolous, we can measure it to 1000. Why use such a misleading number, of course you will get a conclusion that is way off.I have still not seen a calculation including the whole chain that can explain how a cold atmosphere can heat a warm surface. Back radiation is about photons from single molecules and that is not possible to calculate at this scale. We should stick to what we know works, heat transfer and thermic radiation. That tells us that a body radiaties only according to its own temperature.”So, looking at the atmosphere as a heat sink is faulty reasoning – it is an insulator.”Insulators is a specific thing, it does not have steep gradients, it has very low gradients.”But all you really needed, in order to know that the atmosphere is raising the surface temperature, is to do the calculations for the blackbody equilibrium temperature for an airless barren planet at 1 AU around the Sun. Since that is lower than what we've got here, it is a clear indication that the atmosphere is responsible for raising the temp. All that remains is to figure out how (which the paper linked above does nicely).”I think they calculate with average watts, not what is hitting earth in reality, 1370W. That is almost 400K. Look at the moon, above 100C.”The Stefan-Boltzman law concerns only radiative energy transfer from a black body. You can't use it for thermals.”It´s the base for heat transfer, there is no process that can change it in an open system. And earth surface is cooling to space with a ΔT to 3K, the stuff in between is details that can´t change that without a boundary with conduction.And the whole base for greenhouse theory is about radiation, so where does that leave us?And please tell us what to use then. A chart with numbers of Watt is a mockery of thermodynamics without calculations. And the numbers is clearly wrong, the atmosphere is not that warm. Even though you claim S-B is not correct for the transfer, it IS correct for the temperature.”No! The violation would be if there was colder atmosphere heating up hotter surface, whereas what we've got is the hot Sun heating up the surface.When considering the Earth+atmosphere system, you don't get any NET heat flow inward. Heat is always flowing away from the hot source (surface) to colder surroundings (including space). But there is extra energy coming inward from the atmosphere that wouldn't be there without air, which means that the NET heat flow is lower and the equilibrium temperature at the surface has to self-adjust to re-emit that extra energy.”But heat transfer is a process where the energy in the colder body does not influence the hot body in any way. The hot body radiates only according to it´s own temperature, and as you said, that comes from the sun. There is not a single observation in nature or in experiments where we can see such a process.”You should forget everything you wrote about GR there and there on after, since it's completely misappropriated, and just wrong.Forget about the c^2 in the equation, it's confusing you. It's just a unit conversion factor, and you can freely choose units in which it's equal to 1, so that all the equation says is that mass of a body at rest has some associated energy.”Now you just make me more interested, that was a sore toe.”You're mostly talking about energy conservation anyway, which is not violated when putting CO2 into the atmosphere (because the mass was already there, only not in the atmosphere) nor when increasing temperature, because the system is not closed – i.e. the Sun provides energy. If you would design a rather implausibly good insulation system for the planet, you could raise its temperature as high as the temperature of the Sun's surface, and it wouldn't violate any conservation nor thermodynamic laws.”Absolutely not. Give me the calculations. You are ignoring the distance. Please, up the level.”YWhy would CO2 on Earth heat the Sun? It doesn't make any sense..”Because it is a chain of δT^4 that goes only in one direction, and you are putting in energy at the end exhaust.”No, that is a tell-tale sign of a good insulator.This might be another significant issue – what you seem to consider 'cooling' is just heat transfer from hot to cold. Remember that we're talking about systems in thermal equilibrium with their surroundings, which include heat sinks and heat sources. In such a system there will always be temperature gradients, but this doesn't necessarily mean there is cooling”Well then, show me a good insulator in an example that has a steep gradient from a hot surface.”E.g. if you put on a sweater in winter, a steep gradient from your skin to the air will set up. By your usage of the term, the sweater is 'cooling' your skin, because you're loosing heat through it.”You have to buy new sweaters. The sweater is not hot if it keeps you warm.”For something to be cooling a body heated by an external source, it has to increase energy transfer from the body, so that more net heat is removed than without it, and the equilibrium temperature drops as a result.E.g., if you put a radiator on your processor, it'll remove more energy by increasing surface area in contact with air, reducing its temperature – i.e. a cooling effect. Conversely, if you glue a block of fibreglass to the processor, it'll slow down heat transfer, heating it up. The second case will have a steeper temp gradient than the first, but it will be undoubtedly heating.”So, youre saying that the atmosphere is like fiberglass?It really is a big problem with the comparisons using sweaters, blankets and fiberglass. It shows you have a hard time yourself to validate the theory.Can you show me an open system without fiberglass that gets warmer by being in contact with a much cooler gas… that is in contact with an optimal heat sink?The claim has to be confirmed by an observation in natural setting in scale. Why is it not possible to demonstrate in experiments?There is no proof of the concept. You say that a hot body surrounded by 3K is insulated by a lowdensity, low heat capacity gas. Why cant we design that and make it work then?
[QUOTE="haruspex, post: 5517249, member: 334404"]Interception and reradiation back to the source is a slowdown in transfer but not a slowdown in primary emission. You are confusing the two.”While I am very confused about what "re-radiation" is about, a constant emission from the surface is exactly my point. If emission is constant, nothing more is absorbed than what it gets from the sun. A surface emits according to it´s own temperature only, if emission is constant, that is -not slowed down-, then the "re-radiation" has not addedd anything. What you are saying is that the atmosphere gets heated, nothing else. According to Kirchoff, what is emitted is absorbed, so constant emission is a sign of nothing absorbed from the atmosphere.And "re-raditation" only happens if there are two surfaces. Heat and radiation are waves, light is a wave, you cannot interfere a wave in an open system with a gas receiving heat from a hot body without a boundary or surface, right?”As you should know, the temperature gradient is a consequence of the adiabatic cooling as air rises. This puts a brake on convection, requiring a minimum gradient before convection can occur. Yes, the air cools, but not as a result of heat transfer..”I´m not sure about that brake, how do you mean? How big is the restriction? The speed is pretty constant, but how about pressure?”No, it's quite patchy. One reason CO2 is important is that it plugs some holes in the H2O spectrum. Each absorption band is somewhat spread by Doppler and other effects. You may have read that the greenhouse impact of a gas rises logarithmically with concentration. That is because, typically, the main consequence of increased concentration is a broadening of the bands. In the centre of the band absorption is fairly total, so it is the broadening of the band shoulders that is of interest.”Not the whole spectrum, over the whole spectrum. And strongly at shorter wavelenghts. The doppler is at higher altitude, I think. By logartihmically you mean less effect with higher concentration, right? Not the other way around. “I have no idea how you conclude that. Have you considered how Venus has such a high surface temperature?”Yes, it´s a strange thing. Almost no sunlight reaches the surface. But the pressure of 90+ atmospheres and co2 at 63kg per m^3 is more like a solid than a gas, and as far as i know co2 is considered to be a supercritical fluid at the circumstances. Things I don´t know that much about…yet.What really is the interesting question is why venus has almost the same temperature at 1atm pressure, at around 50km altitude, as earth? That is a really good sign that co2 has very little to do with surface temp.”So when you wear a sweater, as opposed to simply a windcheater, it only makes the sweater warm, not you?”No no, a sweater keeps air in place in a structure, and air is a good insulator when it is not allowed to leave with the heat. And it conducts poorly. But look what happens when you get to hot, water takes over wetting your sweater making conduction easier and cools by evaporation.
[QUOTE="rbelli1, post: 5517213, member: 315621"]So you are saying that radiative cooling is impossible? Then how does this work? https://spaceflightsystems.grc.nasa.gov/print/cooling_prt.htm BoB”[QUOTE="rbelli1, post: 5517213, member: 315621"]So you are saying that radiative cooling is impossible? Then how does this work? https://spaceflightsystems.grc.nasa.gov/print/cooling_prt.htm BoB” I think I saw liquid ammonia in there somewhere;)We can design forced cooling systems, we´re really good at it. Radiative cooling is very possible, that is the main cooling function in the universe(I think). In a natural, unforced, heat transfer between two bodies, the rate of transfer changes only at the cooler body, the hotter one doesn´t take notice of what is riding on it´s wave.
Much of that 80 watts of latent heat gets deposited above 80% of the greenhouse gas [ATTACH=full]103078[/ATTACH]where its transport mechanism changes from convection to radiation, both upward and downward of course,[ATTACH=full]103079[/ATTACH]i've not been able to figure whether they model it that wayDownward bound has to get back through the ghg layer., upward doesn'tfrom last page”Thus, the downwellingLW flux exists as one of the principle uncertainties inthe global surface energy budget. (page 6)In our analysis, the biggest uncertainty and biascomes from the downward longwave radiation. Thissource of uncertainty is likely mainly from clouds. (page 10)”old jim
Sorry for the late response to my part of the conversation. I didn't want to make this a wall of quotes (and kinda hoped haruspex would take over my bit as well), but I couldn't figure out any other way to go about it. I'll try to summarise the crux of the issue as what I think it is first, and then address some of the particular misconceptions in the spoiler.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]Of course all energy leaves as radiation and it leaves from the top of atmosphere. As far as I can see it doesn´t matter that the gas is distributing the heat by cooling the surface at any location to all the volume of the atmosphere. That is a process that maximize the atmospheres capacity to radiate to space.[/quote]The last sentence here seems to be the culprit here. You're seeing the atmosphere as an additional heat sink – i.e., the idea seems to be that since for a set amount of incoming radiation Z, surface radiates X energy to space, then if we add a medium that will remove extra Y energy from the surface, and carry it away where it will then escape into space, it should mean that we've added another sink, so that the energy escaping is X+Y, meaning the radiative emissions have to go down, meaning the equilibrium temperature at the surface must become lower.The issues with this picture are:- the atmosphere is for the most part not transparent to the outgoing radiation, so it can't just escape into space – it gets absorbed and reradiated in all directions, including downwards. The actual atmospheric window for escaping radiation is just about 40 W/m^2.- the thermal heat transfer (conduction, convection, evaporation) from the surface is 1. small when compared with radiative transfer, and 2. ends up being reradiated in upper parts of the atmosphere, again including back to the surface.This could still be a valid objection if the energy balance at the surface was a net removal of energy, and would require quantifying – if it hasn't been done many times already. E.g. see the following paper (with its inforgraphic reproduced below):http://journals.ametsoc.org/doi/pdf/10.1175/2008BAMS2634.1 All that energy that was removed from the surface and then returned in the form of back radiation changes the energy balance at the surface so that there is more incoming energy Z, and the equilibrium has to change to increase radiation (or thermals, but their magnitude is secondary), meaning increase in temperature of the surface.So, looking at the atmosphere as a heat sink is faulty reasoning – it is an insulator.But all you really needed, in order to know that the atmosphere is raising the surface temperature, is to do the calculations for the blackbody equilibrium temperature for an airless barren planet at 1 AU around the Sun. Since that is lower than what we've got here, it is a clear indication that the atmosphere is responsible for raising the temp. All that remains is to figure out how (which the paper linked above does nicely).I'll put the rest into spoilers, since it's all a bit tangent to the main issue, I believe.[spoiler][quote]Black body radiation is a beutiful concept and S-B law is the reason that we can use space as a heat sink. Because it defines the energy leaving the boundary of a system that transfers heat to the surroundings as an effect only depending on temperature. It is true for the transfer of heat no matter if it is in the state of radiation or heat as kinetic energy. It is used for heat transfer and thermic radiation.”The Stefan-Boltzman law concerns only radiative energy transfer from a black body. You can't use it for thermals.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]According to theory of heat transfer, the rate of transfer is defined by the difference in temperature, and it shrinks as the difference gets smaller. So that makes a transfer of energy from atmosphere to surface a violation of the concept black body and even more for grey body.”No! The violation would be if there was colder atmosphere heating up hotter surface, whereas what we've got is the hot Sun heating up the surface.When considering the Earth+atmosphere system, you don't get any NET heat flow inward. Heat is always flowing away from the hot source (surface) to colder surroundings (including space). But there is extra energy coming inward from the atmosphere that wouldn't be there without air, which means that the NET heat flow is lower and the equilibrium temperature at the surface has to self-adjust to re-emit that extra energy.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]Another interesting thing is that I always had a hard time visualizing Einsteins theory of relativity, E=mc^2, but the greenhouse theory solved that for me.”You should forget everything you wrote about GR there and there on after, since it's completely misappropriated, and just wrong.Forget about the c^2 in the equation, it's confusing you. It's just a unit conversion factor, and you can freely choose units in which it's equal to 1, so that all the equation says is that mass of a body at rest has some associated energy.You're mostly talking about energy conservation anyway, which is not violated when putting CO2 into the atmosphere (because the mass was already there, only not in the atmosphere) nor when increasing temperature, because the system is not closed – i.e. the Sun provides energy. If you would design a rather implausibly good insulation system for the planet, you could raise its temperature as high as the temperature of the Sun's surface, and it wouldn't violate any conservation nor thermodynamic laws.And yes, that means that a hotter Earth has more energy stored, i.e., is more 'massive', i.e., curves space-time more.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]And a calculation of radiation balance at any point has to relate to the source, the sun. I f co2 heats the surface as the last passive body in the chain, then it has to heat the sun as well if it is true.”Why would CO2 on Earth heat the Sun? It doesn't make any sense.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]That is also confirmed by observation of energy as temperature in the atmosphere, steep gradients an very low temperatures that is averaged much below surface, is the tell-tale sign of something acting cooling on the surface.”No, that is a tell-tale sign of a good insulator.This might be another significant issue – what you seem to consider 'cooling' is just heat transfer from hot to cold. Remember that we're talking about systems in thermal equilibrium with their surroundings, which include heat sinks and heat sources. In such a system there will always be temperature gradients, but this doesn't necessarily mean there is cooling.E.g. if you put on a sweater in winter, a steep gradient from your skin to the air will set up. By your usage of the term, the sweater is 'cooling' your skin, because you're loosing heat through it.For something to be cooling a body heated by an external source, it has to increase energy transfer from the body, so that more net heat is removed than without it, and the equilibrium temperature drops as a result.E.g., if you put a radiator on your processor, it'll remove more energy by increasing surface area in contact with air, reducing its temperature – i.e. a cooling effect. Conversely, if you glue a block of fibreglass to the processor, it'll slow down heat transfer, heating it up. The second case will have a steeper temp gradient than the first, but it will be undoubtedly heating.I might have skipped a few points, but this response is already way too bloated.[/spoiler]
All that energy that was removed from the surface and then returned in the form of back radiation changes the energy balance at the surface so that there is more incoming energy Z, and the equilibrium has to change to increase radiation (or thermals, but their magnitude is secondary), meaning increase in temperature of the surface.So, looking at the atmosphere as a heat sink is faulty reasoning – it is an insulator.But all you really needed, in order to know that the atmosphere is raising the surface temperature, is to do the calculations for the blackbody equilibrium temperature for an airless barren planet at 1 AU around the Sun. Since that is lower than what we've got here, it is a clear indication that the atmosphere is responsible for raising the temp. All that remains is to figure out how (which the paper linked above does nicely).I'll put the rest into spoilers, since it's all a bit tangent to the main issue, I believe.[spoiler][quote]Black body radiation is a beutiful concept and S-B law is the reason that we can use space as a heat sink. Because it defines the energy leaving the boundary of a system that transfers heat to the surroundings as an effect only depending on temperature. It is true for the transfer of heat no matter if it is in the state of radiation or heat as kinetic energy. It is used for heat transfer and thermic radiation.”The Stefan-Boltzman law concerns only radiative energy transfer from a black body. You can't use it for thermals.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]According to theory of heat transfer, the rate of transfer is defined by the difference in temperature, and it shrinks as the difference gets smaller. So that makes a transfer of energy from atmosphere to surface a violation of the concept black body and even more for grey body.”No! The violation would be if there was colder atmosphere heating up hotter surface, whereas what we've got is the hot Sun heating up the surface.When considering the Earth+atmosphere system, you don't get any NET heat flow inward. Heat is always flowing away from the hot source (surface) to colder surroundings (including space). But there is extra energy coming inward from the atmosphere that wouldn't be there without air, which means that the NET heat flow is lower and the equilibrium temperature at the surface has to self-adjust to re-emit that extra energy.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]Another interesting thing is that I always had a hard time visualizing Einsteins theory of relativity, E=mc^2, but the greenhouse theory solved that for me.”You should forget everything you wrote about GR there and there on after, since it's completely misappropriated, and just wrong.Forget about the c^2 in the equation, it's confusing you. It's just a unit conversion factor, and you can freely choose units in which it's equal to 1, so that all the equation says is that mass of a body at rest has some associated energy.You're mostly talking about energy conservation anyway, which is not violated when putting CO2 into the atmosphere (because the mass was already there, only not in the atmosphere) nor when increasing temperature, because the system is not closed – i.e. the Sun provides energy. If you would design a rather implausibly good insulation system for the planet, you could raise its temperature as high as the temperature of the Sun's surface, and it wouldn't violate any conservation nor thermodynamic laws.And yes, that means that a hotter Earth has more energy stored, i.e., is more 'massive', i.e., curves space-time more.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]And a calculation of radiation balance at any point has to relate to the source, the sun. I f co2 heats the surface as the last passive body in the chain, then it has to heat the sun as well if it is true.”Why would CO2 on Earth heat the Sun? It doesn't make any sense.[QUOTE="Reality Is Fake, post: 5516436, member: 598755"]That is also confirmed by observation of energy as temperature in the atmosphere, steep gradients an very low temperatures that is averaged much below surface, is the tell-tale sign of something acting cooling on the surface.”No, that is a tell-tale sign of a good insulator.This might be another significant issue – what you seem to consider 'cooling' is just heat transfer from hot to cold. Remember that we're talking about systems in thermal equilibrium with their surroundings, which include heat sinks and heat sources. In such a system there will always be temperature gradients, but this doesn't necessarily mean there is cooling.E.g. if you put on a sweater in winter, a steep gradient from your skin to the air will set up. By your usage of the term, the sweater is 'cooling' your skin, because you're loosing heat through it.For something to be cooling a body heated by an external source, it has to increase energy transfer from the body, so that more net heat is removed than without it, and the equilibrium temperature drops as a result.E.g., if you put a radiator on your processor, it'll remove more energy by increasing surface area in contact with air, reducing its temperature – i.e. a cooling effect. Conversely, if you glue a block of fibreglass to the processor, it'll slow down heat transfer, heating it up. The second case will have a steeper temp gradient than the first, but it will be undoubtedly heating.I might have skipped a few points, but this response is already way too bloated.[/spoiler]
[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]slowdown in heat transfer is something that only happens at the receiving end. The source don´t care about what is licking up the waste it´s putting out in radiation.”Interception and reradiation back to the source is a slowdown in transfer but not a slowdown in primary emission. You are confusing the two.[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]The air cools so fast with height that it´s clear that there is a cooling effect all the way up there.”As you should know, the temperature gradient is a consequence of the adiabatic cooling as air rises. This puts a brake on convection, requiring a minimum gradient before convection can occur. Yes, the air cools, but not as a result of heat transfer.[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]co2 and water absorbs over the whole spectrum from 700nm to way up in far-IR.”No, it's quite patchy. One reason CO2 is important is that it plugs some holes in the H2O spectrum. Each absorption band is somewhat spread by Doppler and other effects. You may have read that the greenhouse impact of a gas rises logarithmically with concentration. That is because, typically, the main consequence of increased concentration is a broadening of the bands. In the centre of the band absorption is fairly total, so it is the broadening of the band shoulders that is of interest.[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]From S-B law applied in heat transfer and thermic radiation it is clear that there is no point in adressing the re-emission from a certain gas at this scale in an open system”I have no idea how you conclude that. Have you considered how Venus has such a high surface temperature?[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]A warming would not show at the surface, it would show at the other end of transfer. At the tropopause”So when you wear a sweater, as opposed to simply a windcheater, it only makes the sweater warm, not you?
[QUOTE="Reality Is Fake, post: 5516725, member: 598755"]A slowdown in heat transfer is something that only happens at the receiving end. The source don´t care about what is licking up the waste it´s putting out in radiation. It is only cooled when another way of transfer is added to radiation, like conduction or convection.”So you are saying that radiative cooling is impossible?Then how does this work? https://spaceflightsystems.grc.nasa.gov/print/cooling_prt.htmBoB
[QUOTE="haruspex, post: 5516530, member: 334404"]Only up to the tropopause. Going any further depends almost entirely on radiation.”But it does it really well all the way up to the tropopause. From about 300K down to about 200K in 10km. The air cools so fast with height that it´s clear that there is a cooling effect all the way up there. Observed temperatures shows a cooling in the troposphere at any altitude. I can´t figure out how we can interprete such a steep gradient as anything but poweful cooling. That type of gradient is what we want when we design a device that cools by convection, why would a troposphere that shows such a gradient be a heating element?And wher it´s hotter than else, we see a higher troposphere, at the equator. So there seems to be a regulating mechanism that keeps the temperature constantly at the same level at tropopause. Funny enough, co2 shows it´s imprint in spectral intensity of OLR at the same temperature as the tropopause. The sign of co2 in spectral is that of a massive reduction in intensity att the peak wavelength of surface radiation at 300K.I feel like someone is making fun of me, all the signs that we observe in the atmosphere, and from co2 especially, is the signs of cooling. From earth to space. But everyone says it´s warming. Some even point to that decrease in intensity of radiation from co2 distinct absorption at 15μ. As far as I know, an increase in temperature must show as an increase in total intensity. The argument that it shows trapped heat doesn´t add up. Heat doesn´t hide.[QUOTE="haruspex, post: 5516530, member: 334404"]It depends what you mean by "trapping". Infrared Radiation is absorbed from either direction (up or down) and reradiated equally in both directions. Since the Earth reradiates absorbed visible and IR as IR, there is more IR coming from below than from above. The net effect of the greenhouse gas is therefore to slow the transfer of heat into space. That is all that is meant by trapping in this context.”Yeah, I know, I´ve seen that explanation everywhere. But mostly with the addition that it heats the surface as well. I´m totally agreeing with earth heating the atmosphere, that is clear. And co2 and water absorbs over the whole spectrum from 700nm to way up in far-IR. With much more intense radiation in near-IR. I have not managed to clarify how many watts the atmosphere absorbs in near-IR compared to far-IR. But it looks like about the same, maybe more in the shorter wavelenghts from insolation.From S-B law applied in heat transfer and thermic radiation it is clear that there is no point in adressing the re-emission from a certain gas at this scale in an open system. We get the best result when we consider difference in temperature as the rate of transfer. And when that rate is calculated we cannot se any change the rate of emission or the temperature at the warmer surface. And I think that is supported by observation as well. A warming would not show at the surface, it would show at the other end of transfer. At the tropopause, since all transfer of heat to other altitudes below must be less. Even if the atmosphere is slowing the rate of transfer into space, that is only from the atmosphere. The surface emits only according to it´s own temperature, and of course that is according to the suns temperature.Some people seems to approach this "trapping" of radiation in the atmosphere as resistance in an elecrical circuit, or as a resistance in the flow. While it´s not totally wrong, it doesn´t say anything about the surface temperature. Electrical circuit resistance does not heat the source, it heats the resisting medium for the flow. A resistance in a flow is more like a stone in a waterfall, it doesn´t change the flow at the source.One more point, in heat transfer, an atmospheric gasmixture at 1 atm of pressure, it is said that for a temperature of 300K radiation is absorbed, and for higher temps, like 1000K, it is absorbed and emitted. I think that it is not until we see the spectral sign of co2 emitting radiation, which would be seen as a rising spike, that we can come to the conclusion that we are observing a rise in temperature that is signed by co2. It should also show up as a smaller gradient and/or a higher overall troposphere.
[QUOTE="haruspex, post: 5516640, member: 334404"]Below that, convection carries the heat up.”A lot of it by water vapor.If this number is goodhttp://www.superstrate.net/pv/illumination/irradiation.html“The solar energy irradiated to the Earth is 5.10[SUP]24[/SUP] Joule per year.”it would seem that in short-circuiting the insulating layer, hurricanes and thunderstorms provide a goodly part of a 1% energy trim mechanism to keep the tropical oceans cool.
[QUOTE="jim hardy, post: 5516626, member: 327872"]Once air reaches tropopause it's above most of the greenhouse gas so radiation has a shorter hop to make”Sure, but that is the bit where it matters. Below that, convection carries the heat up.
[QUOTE="haruspex, post: 5516530, member: 334404"]The net effect of the greenhouse gas is therefore to slow the transfer of heat into space. That is all that is meant by trapping in this context.”Once air reaches tropopause it's above most of the greenhouse gas so radiation has a shorter hop to makeHurricanes can move something like 10^22 joules a year up therehttp://www.aoml.noaa.gov/hrd/tcfaq/D7.htmlten twenty day storms in a season ? lately we're having more…Is that a significant number ?
[QUOTE="Reality Is Fake, post: 5516452, member: 598755"]convection by rising air. Which is the dominant way of heat transfer from the surface of earth.”Only up to the tropopause. Going any further depends almost entirely on radiation.[QUOTE="Reality Is Fake, post: 5516452, member: 598755"]The concept of stopping heat or thermic radiation towards space, or "trapping" it as often is said, is a nonexistant phenomenon in calculated heat transfer and thermic radiation.”It depends what you mean by "trapping". Infrared Radiation is absorbed from either direction (up or down) and reradiated equally in both directions. Since the Earth reradiates absorbed visible and IR as IR, there is more IR coming from below than from above. The net effect of the greenhouse gas is therefore to slow the transfer of heat into space. That is all that is meant by trapping in this context.
[QUOTE="mheslep, post: 5332308, member: 70823"]Heat capacity of the molecule is no more relevant to radiative forcing than is the heat capacty of glass in a greenhouse. The ability of CO2 to scatter certain infrared wavelengths is the issue, that is, to stop some infrared from escaping to space.”The greenhouse glass is a conductive boundary, preventing heat transfer in other ways than conduction. Primarily by preventing convection by rising air. Which is the dominant way of heat transfer from the surface of earth.Co2 absorbs thermal radiation and acts as a body in heat transfer with the surface. S-B law can be used for that and is very clear about the rate of heat transfer as the difference in temperature as the only regulator of the rate. Two bodys at same temperature does not exchange heat and the rate increases with difference in temperature. There is no "stopping" infrared from escaping to space that is at 3K in vacuum. We know from applied thermodynamics in constructions using S-B law for heat transfer, that the radiation from a surface is dependant only of temperature, and the rate of heat transfer is defined by the difference in temperature only. The concept of stopping heat or thermic radiation towards space, or "trapping" it as often is said, is a nonexistant phenomenon in calculated heat transfer and thermic radiation.
[QUOTE="Bandersnatch, post: 5515065, member: 399360"]Hi [USER=598755]@Reality Is Fake[/USER] :welcome:I'm having a little trouble parsing your post, so correct me if I misunderstood your main point.Space, despite being close to 0K in temperature, when treated as a heat sink, doesn't have an arbitrarily high capacity to remove heat. Since the only mode of heat transport is radiative, you're limited by the black body radiation (i.e., Stefan-Boltzman law). All the effects you mentioned – blowing air over a hot surface or evaporative cooling of water – are internal to the Earth-atmosphere system. They only serve to redistribute heat across the planet, but they can't remove any heat from the system. In the end, everything must leave via radiation.”I´m sorry if it´s hard to understand my writing, english is not my native language.Of course all energy leaves as radiation and it leaves from the top of atmosphere. As far as I can see it doesn´t matter that the gas is distributing the heat by cooling the surface at any location to all the volume of the atmosphere. That is a process that maximize the atmospheres capacity to radiate to space.Black body radiation is a beutiful concept and S-B law is the reason that we can use space as a heat sink. Because it defines the energy leaving the boundary of a system that transfers heat to the surroundings as an effect only depending on temperature. It is true for the transfer of heat no matter if it is in the state of radiation or heat as kinetic energy. It is used for heat transfer and thermic radiation.Are you saying that if we consider an imaginary furnace transferring heat from a heat source to a gasmix like the atmosphere that surrounds the source, then we would need no isolating walls if it was surrounded by zero-degree vacuum?Wouldn´t that make gravity the force preventing cooling?As I see it, earth doesn´t have any internal system above the surface that can be treated as an enclosure. The thing that defines an enclosure is that it has a boundary that is a surface or has surface-like properties that transfer heat by conduction. A boundary must be heated and then emit energy according to it´s temperature, and the transferred heat is dependant on temperature only. According to S-B law = P/A=σT1^4 and for graybodys the temperature for all parts is lowered by emissivity.So, yes, all energy leaves by radiation. And, yes, according to S-B law that is valid for both thermic radiation and heat transfer in gasses, we can calculate the transfer from earth to space as an optimal heat sink. That makes the atmosphere an added value of heat transfer to the surface, since there is no barrier that transfer by conduction at the boundary. And heat transfer is defined by S-B law only as a product of temperature, which makes it clear that the lower temperature of the atmosphere, only acts as a receiver of the energy from the surface. The temperature of the source is constant even if it transfers heat to a colder body, when in an open system without boundary. If we study the internal structure of earth we see the difference to a closed system. Very little heat is transfered at the boundary, the earth surface.According to theory of heat transfer, the rate of transfer is defined by the difference in temperature, and it shrinks as the difference gets smaller. So that makes a transfer of energy from atmosphere to surface a violation of the concept black body and even more for grey body. It only happens in closed systems with boundarys that reflects a large fraction and conducts a small fraction to the surrounding outside. The gas transfers heat back to the source when the gas has reached an even temperature in the system. That is the opposite of the earth surface and it´s atmosphere.”If you then put something in the way of the outgoing radiation, so that part of it gets reflected back to the surface, you end up raising the surface to a higher equilibrium temperature..”Only if the reflected radiation is hotter than the surface, hot as in higher energy radiation. And there is no reflection, the gasses absorb and emit, and that is a product of temperature only. Applying S-B for heat and radiation for absorbing gas gives a rate of heat transfer that adds up to much below 0 for the low atmosphere temperature and earth surface. But we only need to apply it for heat gain in the gas, the earth surface will not gain anything from the low temp.”In you example with the rock by a fire place – if you could modify the experiment to use close-circulation air supply, and ensure that the only way heat can leave the system is via radiation (and keep the radiative surface area the same etc.), then as soon as the system reaches thermal equilibrium you won't get any cooling from the air circulating inside.”And that would be a system that had an totally even temperature in the gas surrounding the heat source, which is thermal equilibrium. The opposite of what we observe in the atmosphere that has a very steep gradient from the surface and the average temperature of the whole volume is a lot less than the surface. According to S-B law there is no doubt that the transfer of heat goes in one direction only and that is towards a bottomless heat sink. The atmosphere is just a big lossy extension of surface area from 2 to 3 dimensions, even 4 dimensions as it spreads heat transfer in time, and it is a large addition to the capacity of transfer in relation to the energy absorbed from insolation by only half the surface of the earth.As I wrote before, I´ve been thinking and reading a lot about this and it really is a problem in the way earth temperature is approached in the greenhouse theory. There is a few things we can be sure of, that the sun can be treated as an almost perfect blackbody, the earth is definatly a grey body, and that makes it clear that the laws for radiation, heat, effect and temperature is the only way that we can define the interaction. Those laws, that is applied in many ways in technology and very functional, says that the emitted energy from a planet like earth heated by the sun, is a product of temperature only. The atmosphere heated by the earth is a product of temperature only. Transfer of heat in the system has a rate that is equal to the difference in temperature and it is lower for a small difference. When temperature is equal in two bodys in contact, there is no transfer of heat between the bodys. Heat is never transferred from cold to hot and that is proven by S-B law that gives the rate of transfer.Another interesting thing is that I always had a hard time visualizing Einsteins theory of relativity, E=mc^2, but the greenhouse theory solved that for me.Since mass * speed of light is equal to energy, and speed of light is the definition of a photon, and earth´s energy that is equal to temperature which is a product of photons radiated from the sun, we can be absolutely sure of that added co2 cannot heat the earth when all other things is constant.If E/m=c^2 and E=Temperature of the earth, then an increased mass in the atmosphere by added co2 without solar insolation increasing, would result in an increased speed of light in the photon if Energy that is divided by mass is increased. Which we know is an absolute limit for speed, as calculated for macroscopic relations in space. This is because the source of energy is external and fixed in addition to c that is an upper limit. The only thing that changes is the mass, and that actually would result in a decrease in energy measured as lower temperature of earth. A very small decrease but still, it seems like the whole theory of an atmosphere heating anything as an added volume of low density mass with a low emissivity above the surface, is a direct violation of E=mc^2. And even more so, added co2 to any calculation cannot change temperature without adding more energy from the source, that is our sun.I have searched intensivly for a complete calculation with S-B law that shows heating from the icecold atmosphere, and have found none. It has to include all bodys, the sun, earth and it´s atmosphere. And a calculation of radiation balance at any point has to relate to the source, the sun. I f co2 heats the surface as the last passive body in the chain, then it has to heat the sun as well if it is true. But it is not necessary to go that far, we only need to apply S-B law for heat transfer that is valid for both kinetic energy and radiation, dependant on temperature only, and there is no question that it is a violation of the relationship between matter and energy.That is also confirmed by observation of energy as temperature in the atmosphere, steep gradients an very low temperatures that is averaged much below surface, is the tell-tale sign of something acting cooling on the surface. I was quite happy when I realized that I finally can visualize Einsteins theory in matter with radiation of photons and energy as temperature of mass.I don´t want to come across as a sceptic of AGW, I´m just really trying to figure out how added mass of co2, without added energy from the source, can raise temperature by heat transfer from hot to cold? And basically, how can an atmosphere that is an added low density volume to the surface, warm the earth?When the numbers add up wihout an atmosphere? And why do we treat water and air as warming in the atmosphere when we never use it in that way in daily life?
[QUOTE="haruspex, post: 5327846, member: 334404"]Thanks D H, I was not aware that was wrong. But I like the image, much clearer than most, so I've just added a note. (Is the curve wrong, or just the number? The peak seems to be in about the same place as in other images I found.)”I think the curve is useful and accurate enough. So I would suggest changing the number on the image, and changing the note to indicate what you did.I see no reason to leave that uncorrected error.
Hi [USER=598755]@Reality Is Fake[/USER] :welcome:I'm having a little trouble parsing your post, so correct me if I misunderstood your main point.Space, despite being close to 0K in temperature, when treated as a heat sink, doesn't have an arbitrarily high capacity to remove heat. Since the only mode of heat transport is radiative, you're limited by the black body radiation (i.e., Stefan-Boltzman law). All the effects you mentioned – blowing air over a hot surface or evaporative cooling of water – are internal to the Earth-atmosphere system. They only serve to redistribute heat across the planet, but they can't remove any heat from the system. In the end, everything must leave via radiation.If you then put something in the way of the outgoing radiation, so that part of it gets reflected back to the surface, you end up raising the surface to a higher equilibrium temperature.In you example with the rock by a fire place – if you could modify the experiment to use close-circulation air supply, and ensure that the only way heat can leave the system is via radiation (and keep the radiative surface area the same etc.), then as soon as the system reaches thermal equilibrium you won't get any cooling from the air circulating inside.
[QUOTE="Bandersnatch, post: 5347132, member: 399360"]Intuition from other, physically similar setups can help you here. Consider what it means to be a good heat insulator.For example, you're an Arctic explorer and it's -70 degrees centigrade outside. Yet, thanks to the many layers of good insulation, your internal body temperature is kept at the healthy 36.6 degrees. That's over a 100 K difference. The indication that the clothes you're wearing are a good insulator, is that it lets very little heat escape, and there's a large gradient of temperature as you get deeper inside.This is the same for Earth's lithosphere, made largely of poorly-conducting silicates – if it were a good heat conductor, you wouldn't get an increase in temperature as you get down the mine – the surface temperature would be instead almost the same as that of the deeper layers. Which would incidentally cause the whole planet to cool very rapidly as that hot surface would radiate heat profusely.Again, this is the same as with warm clothing – if you can feel your body temperature when you're touching the outer layers, then it ain't a good thing to wear in the winter.”Hi, new member here. That sounds as just the right metaphore for earth`s intestines. Similar ways of illustrating the relationship between temperature and surroundings is used for atmosphere by many.I think it is a bit misguiding that analogies for the atmosphere is used so often in explaining the effect on surface temperature. It consists of air and we don´t need metaphores for air.The correct version of the Arctics explorer would then be for the atmosphere: how does his temperature of 300K change when it is immersed in air at about 200K?And as I have done a lot of reading about this lately, the right approach to surface temperature in contact with atmospheric gasses is basicly the same way we think about all warm surfaces in contact with cold air. And of course in the long run evaluate all different mechanisms in air that is involved in the observed distribution of temperature.The most important aspect is that air does not insulate very well when it`s not surrounded by a structure that keeps it in place like we observe in insulation within walls or carefully designed surfaces in walls of furnaces that operate at high temps. The best experience is in engineering that have to use the right methods, because the result has to be a design based on a correct understanding of heat and radiation combined with a somewhat complete knowledge of all included transfers of the energy produced by a hot source.Their approach to heat transfer and air which mostly is an intention to contain or shield from heat/radiation, is that air must be kept in/outside an enclosure with walls that keeps transfer to a minimum. Because atmospheric gases that is allowed to come in contact with a heat source and ireact to heat freely like air in contact with earth surface, will transfer very large amounts of energy away from the heat source as heat transfers to all possible points with a lower temperature.Considering this experience in applied theory of heat transfer and thermal radiation that has been succesfully used in constructing devices with increasing performance, the right "analogy" of surface temperature and atmosphere is a hot external source of intense thermal radiation that heat the surface(mainly) of half the absorbing body wich radiates a fraction of that heat to air that surrounds it, reduced by emitting same amount from twice the area that absorbed it, and the gasses that is much lower in temperature at all distances from the surface is immersed in 0K vacuum with no enclosure or insulating barriers that prevent heat transfer from both the surface an the gas by a wide selection of transfer in the molecules absorption wavelenghts. The transfer can happen in almost all wavelengths in the total spectrum for both bodys that is included, and even a bit more. The many diffrent ways of absorbing provides many ways of transfer that I think can be seen as several "lanes" that prevent traffic jams efficiently. With overlaps that enhaces exchange between molecules and also change of wavelenght.The difference to engineering furnaces or other ways to control temperature that is aimed at making the insulating enclosure as effective as possible when it comes to keep the gasses heat capacity under control to do work. We can instead imagine something quite the opposite, that air or other gasses can, without any restrictions, transfer the maximal amount of heat in all possible ways, to an optimal heat sink that has no resistance. A way to increase heat transfer in a furnace is to add a second surface that absorbs heat and reach the same temperature, this increase surfacecontact with the gas that transfer the heat. Adding an atmosphere with high absorbing capacity to earth surface would be very much like adding more surface that is involved in the transfer of energy to the heat sink in space. And also extending the twodimensional surface into three dimensions. I have turned the atmosphere inside out when trying to find the insulating effect, which would be easy to spot in the gradient. There is not any way to interpret observed temperature and behaviour of the gasmix and water, than a clear display of very efficient heat transfer through the whole spectrum into an optimal heat sink. On top of that, consider the absorbed energy/m^2 that is emitted from double the surface area as only one side is irradiated, and that the atmosphere is reducing the effect at the surface before absorbtion. As icing on the cake the atmosphere transport heat away from irradiated surface and volume to the side that is constant cooling. It´s like a ridicoulus excess of cooling functions and then the biggest is not mentioned yet. The oceans acts very much like an AC with compression and expansion in circulation to the icecaps. The top carries heat with high speed to cooling icy waters and sinks to a compressed slow stream at the bottom back to places that warms.My first thought that made me investigate this was: What happens if i put a stone beside a fire and observe the temperature when,*I pour water over it*I place it in a waterbath that covers a part of the surface* I add some really large pieces of ice both on the dry parts and in the waterbath*Blows compressed air across the surface with a rate that is about the same rate as convection works on earth*Adding absorbing gases Which one is raising temperature/limiting loss?I can´t see that there is such a mechansim present. Not around the stone and not anywhere in the atmosphere. All these influences are the foundation in the greenhouse theory that claims the opposite effect on surface temperature than we observe in all other circumstances when heat is transfered from a source with higher temperature towards all colder surroundings. If we think about how these ways of heat transfer in gasses and water from hot surfaces and spaces in normal situations daily, we never treat air or water as a heat transfer from cold temperature fluid or gas to the hot volume or surface of matter.And in relation to threadtopic, what would the internal distribution look like if there was no sun?Our core is even hotter than the suns surface, rightThe difference in about 1000W/m^2 hitting half of surface, ignoring atmosphere, to a heat sink of 0K without any heat source…. that must be more like Pluto or something?Even with an atmosphere there has to be a difference, am I right?
Bandersnatch, thank you once more for an enlightening response! I’d never have guessed that the outer layer of our planet is such a good insulator (basalt and granite being pretty cool to the touch — but not -237 centrigades cool, for which we have to thank the Sun).
“Intuitively, I still find it hard to understand”
Intuition from other, physically similar setups can help you here. Consider what it means to be a good heat insulator.
For example, you’re an Arctic explorer and it’s -70 degrees centigrade outside. Yet, thanks to the many layers of good insulation, your internal body temperature is kept at the healthy 36.6 degrees. That’s over a 100 K difference. The indication that the clothes you’re wearing are a good insulator, is that it lets very little heat escape, and there’s a large gradient of temperature as you get deeper inside.
This is the same for Earth’s lithosphere, made largely of poorly-conducting silicates – if it were a good heat conductor, you wouldn’t get an increase in temperature as you get down the mine – the surface temperature would be instead almost the same as that of the deeper layers. Which would incidentally cause the whole planet to cool very rapidly as that hot surface would radiate heat profusely.
Again, this is the same as with warm clothing – if you can feel your body temperature when you’re touching the outer layers, then it ain’t a good thing to wear in the winter.
“Do you happen to know how warm (cold) Earth would be if there were no Sun? If 0 Kelvin is the absolute minimum and 287 Kelvin is the current average temperature on Earth’s surface, I wonder how much that would be if we only had Earth’s core as an energy source.”
Putting a precise number on that is beyond my paygrade. What I can do is provide an approximation of the upper bound.
Take the Stefan-Boltzman law:
##P=sigma T^4##
We know that for the current surface temperature of ##T_1=300 K## (the calculations are too simplified to worry about being imprecise), the power received from the sun and reradiated by the surface (approximated as a black body) is ##P_1=1.7*10^{17} W##
The energy estimated to be leaving the surface after being conducted from deeper layers (i.e., the internal energy heating up the surface) each second is ##P_2=4*10^{13}W##
By comparing ##P_1/P_2## we can get the temperature of a black body radiating with power equal to only the internal energy loss (at its current magnitude):
##frac{P_1}{P_2}=frac{T_1^4}{T_2^4}##
##T_2= T_1 sqrt[4](frac{P_2}{P_1})##
You plug in the numbers given above, and get the approximate temperature ##T_2 = 36K## or -237 degrees centigrade.
This in an upper bound, because without the Sun, you’d end up with a planet that is not constantly heated up by it, so it’d cool faster over its life, so by today it’d be losing even less heat that it does now. It’s about right an answer if you were looking for a scenario where the Sun magically disappears today.
The atmosphere would probably not affect that temperature by much – at 36K most of the gasses would liquify or solidify, and the peak radiation would be shifted much farther into the infrared, so any greenhouse effects would be negligible.
Point 5 states that
“Compressing a gas heats it, but won’t keep it hot. If the atmosphere were just a static layer of gases, only heated or cooled by conduction, it would all come to the same temperature.”
Would pure heat conduction really result in the same adiabatic temperature gradient?
Just a thought on clouds and the effect of water vapor
the effect of water vapor on convection is tremendous
with Molecular Weight of just 18 vs air’s ~29 , water vapor makes moist air less dense so it rises,
and its enormous heat of vaporization affects lapse rate causing an awful lot moisture laden air to rise above most of the atmosphere’s CO2
[IMG]https://gregklein.files.wordpress.com/2011/01/pressure_vs_altitude.png[/IMG]
at 5km, around 16,000 ft, it’s already above nearly half the atmosphere
and any decent thunderstorm has tops above 20,000 ft
hurricanes more like 50,000
Given that a hurricane removes something like 5 X 10^19 joules per day from the ocean ([URL]http://www.aoml.noaa.gov/hrd/tcfaq/D7.html[/URL]),
”
Subject: D7) How much energy does a hurricane release?
Contributed by Chris Landsea (NHC)
Hurricanes can be thought of, to a first approximation, as a heat engine; obtaining its heat input from the warm, humid air over the tropical ocean, and releasing this heat through the condensation of water vapor into water droplets in deep thunderstorms of the eyewall and rainbands, then giving off a cold exhaust in the upper levels of the troposphere (~12 km/8 mi up)…..
….
Method 1) – Total energy released through cloud/rain formation:
An average hurricane produces 1.5 cm/day (0.6 inches/day) of rain inside a circle of radius 665 km (360 n.mi) (Gray 1981). (More rain falls in the inner portion of hurricane around the eyewall, less in the outer rainbands.) Converting this to a volume of rain gives 2.1 x 1016 cm3/day. A cubic cm of rain weighs 1 gm. Using the latent heat of condensation, this amount of rain produced gives
5.2 x 10[SUP]19[/SUP] Joules/day or
6.0 x 10[SUP]14[/SUP] Watts.
”
If we just USWAG 70 hurricane days a year , ten storms at a week apiece,
that’s 3.64 X 10 [SUP]21[/SUP] joules
comparable to the heat content of earth’s whole inventory of air.
[ATTACH=full]93970[/ATTACH]
[URL]https://www.quora.com/What-is-the-total-heat-content-of-the-Earths-atmosphere-partitioned-between-its-various-layers[/URL]
if this guy is in the ball park
[URL]http://www.ecd.bnl.gov/steve/pubs/HeatCapacity.pdf[/URL]
[ATTACH=full]93971[/ATTACH]
16.7 watt years per square meter per degree
how many joules in that many watt-years ?
16.7 watts = 16.7 Joules/sec, X 60sec/min X 1440min/day X365.25 day/year = 5.27 X 10[SUP]8[/SUP] joules /year X 1 year
multiplied by earth’s area of pi X 12,742000^[SUP]2[/SUP], 5.1 X 10 [SUP]14[/SUP]m[SUP]2[/SUP] = 2.69 X 10[SUP]23[/SUP] joules
our 3.64 X 10[SUP]21[/SUP] joules lifted by hurricanes is only 1.35 % of that number
aha
Sooo can i suggest
hurricanes don’t cool the whole climate system very much
they just help convey heat from the tropics to the upper atmosphere
where it migrates to the poles and radiates away ?
While that amount of heat isn’t very significant to the oceans
it’s darn significant to the air, amounting to 73% of its heat capacity number
and that air is responsible for most of the thermal radiation
[QUOTE=http://earthobservatory.nasa.gov/Features/EnergyBalance/page4.php] The atmosphere radiates heat equivalent to 59 percent of incoming sunlight; the surface radiates only 12 percent. In other words, most solar heating happens at the surface, while most radiative cooling happens in the atmosphere. How does this reshuffling of energy between the surface and atmosphere happen? ”
I wish i were smart enough to work this in my head and ascribe numbers.
Thanks for reading ,
old jim
“Now hundreds or years of accumuated soot are being exposed – that drops the albedo from about 0.9 to 0.1 so a nine fold increase in the solar heating occurs – your will be hard pressed to find any more strong positive feed back than that….”
I don’t think soot (black carbon) is properly described as a feedback of any kind. Rather, soot acts independently of CO2 radiative effects, i.e. a direct driver, like methane or aerosols. That is, if more soot were sprayed on the Greenland ice sheet from, say, a volcanic eruption, the albedo would further decrease even if CO2 concentration suddenly reverted to preindustrial levels.
Forcings graphic from AR4:
[IMG]https://www.ipcc.ch/publications_and_data/ar4/syr/en/fig/figure-2-4-l.png[/IMG]
“The polar regions still have a low “average angle of incidence of insolation” (which is what I thought I said)….”
Yes, but ranging up to 24 hours a day in season.
“Brewster’s angle (53º) is the angle where about half of light is reflected off the surface of water. ”
That is not what Brewster’s angle is. Brewster’s angle is the angle at which none of the p-polarized light is reflected. All of that light enters the water. Most of the s-polarized light also enters the water at this angle. At Brewster’s angle, over 95% of the incident light on a flat air/water surface enters the water. Water is darker than fresh asphalt at this angle. Water is darker than fresh asphalt at most angles. It’s not until the light rays become very close to parallel to the surface (84 degree angle of incidence) that more light is reflected than transmitted.
“Beause it is. The greater heat absorbed by lower albedo melts more ice and snow. lowering the albedo even more as bare (darker) spots develope.
Also often there develope pools of water in low spots and the albedo there is lowered too, compared to the ice under the bottom of the pool. Again, I challenge you to find any Postive Feedback as strong as the one happening NOW in Greenland’s ice sheet.”
While this is likely true for Greenland, it’s not at all clear it’s true for sea ice.
Brewster’s angle (53º) is the angle where about half of light is reflected off the surface of water. This means that above 53º latitude, naively, the albedo of water is over 0.5. (It is far more complicated than that of course — which is my point.) Since ice and snow’s albedo ranges from 0.9 to 0.3, it is not clear water’s albedo is lower than ice in the arctic — even in the summer. (In the winter the albedo is irrelevant because there is effectively no incident sunlight.)
Meanwhile, the arctic area can still “see” the night sky at a 90º angle. Its emissivity remains and the area continues to emit longwave infrared radiation. The limitting factor on this is likely the ∆T[SUP]4[/SUP]between the ground/sea and the night sky. Raising the temperature in the region raises the ∆T and gets that 4[SUP]th[/SUP] power emission bonus.
I want to make it clear I am not claiming this melting is a good thing. I’m just claiming it has been poorly studied. The arctic environment is probably the most fragile in the world. Given the large methane deposits in the tundra and oceans (clathrates) which even small temperature rises might release, there is cause for concern.
I can think of a number of things wrong with my model, the most obvious is that water (and arctic ice for that matter) is not flat. Waves will create a chop effect limiting the albedo gain due to the Brewster angle. The Brewster angle will change with the seasonal axial tilt. The atmosphere above the ice will have a large effect since waves incident at a low angle will travel through much more air. These conundrums are just off the top of my head. My point is that this is a complex subject and any positive feedback loop is at best not obvious.
But the idea that the albedo is of great concern in a region getting little sunlight seems oddly immune to logic. Global warming is a big enough problem without adherents practicing bad science and settling for confirming their bias.
“If the changing polar albedo effect is well understood, why do educated climatologists continually refer to a lowered polar albedo as absorbing more heat as a positive feedback? … “Beause it is. The greater heat absorbed by lower albedo melts more ice and snow. lowering the albedo even more as bare (darker) spots develope.
Also often there develope pools of water in low spots and the albedo there is lowered too, compared to the ice under the bottom of the pool.
It is even more comlex as for hundreds of years (or more) soot from forest fires has been falling on the snow but is soon covered when the snow is not melting, as it is today. Now hundreds or years of accumuated soot are being exposed – that drops the albedo from about 0.9 to 0.1 so a nine fold increase in the solar heating occurs – your will be hard pressed to find any more strong positive feed back than that.
This video discusses the “lakes” forming on the ice increasing solar absorption: [URL]http://cdn.rollingstone.com/feature/greenland/video/lakes.webm[/URL]
If you watch this UCLA video / discusion below, not only will you better understand the positive feed back of melt water effects but also as their sensor moves over the ice, you will see it is quite dark in some areas with decades of accumulated soot are now exposed on the surface.
[MEDIA=youtube]-EMCxE1v22I[/MEDIA] Even without opening this video, you can see the ice/ snow surface is far from the white you expect for snow. Again, I challenge you to find any Postive Feedback as strong as the one happening NOW in Greenland’s ice sheet.
“Water has a 4 times higher heat capacity than CO[SIZE=3]2[/SIZE]. …”I doubt that as H2O is not a linear molecule as 0-C-O is. Thus with an extra non-zero monent of inertia for storing energy in rotation H2O vapor should have slightly greater means of storing energy when heated say, 0.1 C degree.
Water has the two Hs on the same side of the O, with 105 degree angle between them and having partially lost their electron are with positive charged and the O is negatively charged. – Why water molecule has a permanet dipole and is such a powerful radiator and absorber IR compared to CO2 or more graphically, 0-C-O.
“Water has a 4 times higher heat capacity than CO[SIZE=3]2[/SIZE]. Also the heat capacity of air is higher than of CO[SIZE=3]2[/SIZE]. .”Heat capacity of the molecule is no more relevant to radiative forcing than is the heat capacty of glass in a greenhouse. The ability of CO2 to scatter certain infrared wavelengths is the issue, that is, to stop some infrared from escaping to space.
Water has a 4 times higher heat capacity than CO[SIZE=3]2[/SIZE]. Also the heat capacity of air is higher than of CO[SIZE=3]2[/SIZE]. Similar is the course of the conductivity of heat with the lowest amount by CO[SIZE=3]2[/SIZE] (Datas of the Handbook of Chemistry and Physics 14 -24, 81. Ed., 2000/2001). There is a wide range of overlapping of the absorption of the infrared between water and CO[SIZE=3]2[/SIZE]. Mostly in the atmosphere is a higher concentration of water than of CO[SIZE=3]2[/SIZE]. Therefore water should have a higher radiation force than CO[SIZE=3]2[/SIZE]. By most processes, where humans produce CO[SIZE=3]2[/SIZE] and many others much water gets in the atmosphere. The contribution of water of the greenhouse effect is about 60%. Therefor is to avoid the production of steam and heat in first line and not 0,04 % of CO[SIZE=3]2[/SIZE].
Jeff Rosenbury (post 29) said:
“Meanwhile heat energy radiates more or less uniformly.” Not true, not even close. In order for this to be true, the two major radiators, the Earth’s surface and the greenhouse gases would have to be almost isothermal. They are far from that. In actuality, both the wavelengths and the intensities of global terrestrial radiation vary tremendously from place to place and from time to time.
“This causes the poles to be cooler than the equator.” Not true. The poles are cooler because the average angle of incidence of insolation is lower. This, in turn, is due to the fact that the Earth is a sphere.
” . . . actually increases the blackbody radiation . . .” The Earth emits no blackbody radiation. The Earth emits as a greybody. Many scholars put the coefficient of emissivity at about 0.95.
” . . .to get a poorly understood mix of feedback loops.” It is actually fairly well understood, and whole library shelves of books and tens of thousands of scholarly articles have been written on the subject. I refer you to almost any Climatology 101 course.
I, too, have been to the Arctic (Thule, Greenland)–although during the summer months. We enjoyed continuous daylight.
“O2, N2,NO2, & argon all supply “bound electrons” for the incident sunlight’s E-field to shake.”
Hi [USER=536963]@BillyT[/USER]:
Thanks for your post answering my question. I apologize for misinterpreting your
[INDENT]”EM wave is passing thur a dense medium the E-field will oscillate the bound electrons, and an accelerated electron produces radiation which combines with the incident radiation to change the net phase of radiation field.”[/INDENT]
The technical aspects of E-field and change to the net phase of the radiation field are a bit over my head. In the context of the other quote, I was confused and thought you were saying the Einstein / Smoluchowski theory was a sufficient explanation.
Regards,
Buzz
“I wonder how all this figures in climate models.”
This is a negligible effect – while it gets very hot deep inside the planet, this heat is not well conducted towards the surface, and contributes only a minuscule part of the heat the surface receives from the sun.
See here:
[URL]https://en.wikipedia.org/wiki/Earth’s_internal_heat_budget[/URL]
“I read the third section above, “Black Body Earth”, which speaks of an -18 degree equilibrium, but I don’t think I understand. Does that or does that not account for this heat from within the planet? I don’t think it does, or else there wouldn’t be places on Earth where it gets even colder in Winter. Or, maybe those colder places can be explained by regional differences in heat conductivity of the planet’s mantle and crust?”
The equilibrium temperature is the uniform temperature the whole surface would need to have in order to reradiate the specified amount of energy. You can have places during winter, at high latitudes, or simply at night with lower temperatures, as long as there are places with higher temperatures elsewhere.
I.e., it has to be compared with the averaged global temperature, not with specific individual instances.
“Hi [USER=536963]@BillyT[/USER]:
Thanks very much for the reference. I am a bit confused by your quote relative to what I read in the reference.
The quote above seems to be saying that the Einstein / Smoluchowski theory about fluxuations explains the blue sky. The following is a quote from the reference.
[INDENT]Einstein stated that “it is remarkable that our theory does not make direct use of the assumption of the discrete distribution of matter” . . .
There is no dispute, then, about what really causes the sky to be blue? Is it “really” scattering by molecules? Or, if you want to be more precise, scattering by electrons. Even more precise, scattering by bound electrons: free electrons would not give us a blue sky. The preposition “by” indicates an agent, and molecular scattering is the agent responsible for the sky’s blueness.[/INDENT]
This seems to contradict your quote’s suggestion that molecules are [U]not[/U] responsible for the blue sky. Your quote seems to support the Einstein / Smoluchowski theory, and the references seems to say it’s wrong. Or have I misinterpreted what you said? Or what the article says?
ADDED
Now that I have read the whole article, I get that any molecule of sufficiently small size, e.g. argon, would also make the sky blue, provided such molecules have no specific energy levels in its spectrum that would absorb blue light. So the reason that NO2 makes the sky blue is that NO2 makes up most (~80%) of the atmosphere. Since O2 makes up most of the remainder, I am curious. Does O2 also scatter light with smaller wavelengths like blue and violet? Regards, Buzz”Note I said:
“EM wave is passing thur a dense medium the E-field will oscillate the bound electrons, and an accelerated electron produces radiation which combines with the incident radiation to change the net phase of radiation field. (Why the refractive index exist.*) ”
So yes there must be some matter (especially bound electrons) to make a refractive index. O2, N2,NO2, & argon all supply “bound electrons” for the incident sunlight’s E-field to shake.
If the bound electronic density were exactly the same every where, so would be the index of refration and there would be only forward (and bckward) scattering. What Einstein / Smoluchowski theory does is to note that all you need is variation of the index of refraction on wave lenght scale. – you don’t really need to get into the facts relating to how the index of refraction is created, but that was the approach used to explain blue sky originally. Einstein / Smoluchowski skipped the real details about atoms having bound electrons, but they must be present to make the index of refracion, and if their density is varying on the wave length scale, then so will th index of refraction. – One is a high-level POV and the standard, older one is a more detailed POV, but they are essentially the same.
Sort of like the difference between: The IC motor makes torque that turns the wheels vs The vaporized gasoline volume and presure greatly increases as it oxidizes and they forces the pistons down …”
“That gives the Einstein / Smoluchowski theory which does not even require discrete molecules – only density fluxuation, that cause refractive index spatial variation.”
Hi [USER=536963]@BillyT[/USER]:
Thanks very much for the reference. I am a bit confused by your quote relative to what I read in the reference.
The quote above seems to be saying that the Einstein / Smoluchowski theory about fluxuations explains the blue sky. The following is a quote from the reference.
[INDENT]Einstein stated that “it is remarkable that our theory does not make direct use of the assumption of the discrete distribution of matter” . . .
There is no dispute, then, about what really causes the sky to be blue? Is it “really” scattering by molecules? Or, if you want to be more precise, scattering by electrons. Even more precise, scattering by bound electrons: free electrons would not give us a blue sky. The preposition “by” indicates an agent, and molecular scattering is the agent responsible for the sky’s blueness.[/INDENT]
This seems to contradict your quote’s suggestion that molecules are [U]not[/U] responsible for the blue sky. Your quote seems to support the Einstein / Smoluchowski theory, and the references seems to say it’s wrong. Or have I misinterpreted what you said? Or what the article says?
ADDED
Now that I have read the whole article, I get that any molecule of sufficiently small size, e.g. argon, would also make the sky blue, provided such molecules have no specific energy levels in its spectrum that would absorb blue light. So the reason that NO2 makes the sky blue is that NO2 makes up most (~80%) of the atmosphere. Since O2 makes up most of the remainder, I am curious. Does O2 also scatter light with smaller wavelengths like blue and violet?
Regards,
Buzz
“Hi BillyT: Thanks for your post. I am always interested in learning something new. Can you recommend a reference that explains more about this phenomenon? Regards, Buzz”Read the last paragraph of right column, page 269 here:
[URL]http://homepages.wmich.edu/~korista/colors_of_the_sky-Bohren_Fraser.pdf[/URL] That gives the Einstein / Smoluchowski theory which does not even require discrete molecules – only density fluxuation, that cause refractive index spatial variation.
Prior to that Einstein / Smoluchowski theory, the scatering of molecules was used (and still usually is) to explain why the sky is blue. Basically if a plane EM wave is passing thur a dense medium the E-field will oscillate the bound electrons, and an accelerated electron produces radiation which combines with the incident radiation to change the net phase of radiation field. (Why the refractive index exist.*) This electronic radiation goes in all direction in a plain normal to the oscillator motion, but that of many electrons all driven in phase (dense medium & plain wave) only constructively interfer in the forward (or backward, but 180 scatter is weak) direction. I.e. in dense medium of uniform density there is no scattering, but add dynamic density variation on the scale of the wave length you do get scattering.
These small scale density fluxuation are percentage wise much larger in a rarified gas. So most of the scattering of blue light takes place high up in the atmosphere, but not so high that there is little there. Lower in the armosphere, most of the scattering is by small particles (dust etc.).
* As the earlier theory which does consider the individual atomic / molecular scattering gives almost the correct results, few of the papers found by Google have much to say about Einstein / Smoluchowski theory, but it clearly does not depend upon the existance of atoms or molecules – Certainly not what they are. Why I could say that an argon amosphere would also make the sky blue.
“I think that is miss leading. Even if the atmosphere were argon, the sky would still be blue.”
Hi BillyT:
Thanks for your post. I am always interested in learning something new. Can you recommend a reference that explains more about this phenomenon?
Regards,
Buzz
Haruspex,
Like you, I much admire the 1997 paper by Kiehl and Trenberth. I think it should be in every atmospheric scholar’s sourcebook. I believe it to be the standard to which all subsequent global heat budget studies should be compared. Which brings me to another quibble:
In your point 7, you object to the statement, “CO2 is insignificant compared with H2O as a greenhouse gas”. I would object right along with you, but I don’t know any serious scholars of the atmosphere who would make such an unsupported statement. On the other hand, a good many atmospheric scholars would strongly agree that it is a less significant greenhouse gas. Kiehl and Trenberth [1997] are among them. They call the role of water vapor “dominant”.
1. Water vapor accounts for more than 40 Wm[SUP]-2[/SUP] of absorption of short-wave (solar) radiation. Carbon-dioxide accounts for less than 1 Wm[SUP]-2[/SUP] (none, under cloudy skies).
2. The vast majority of photons emitted by terrestrial IR sources are emitted by water molecules. Atmospheric water molecules can absorb all of these photons, atmospheric carbon-dioxide can absorb only a small fraction (the “overlap” wavelengths). Carbon-dioxide plays no significant role in the emission of terrestrial IR.
3. At 4,000 ppm compared to 400 ppm, water vapor is ten times more abundant in our atmosphere than is carbon-dioxide. In your argument you state, “Increasing the level of a relatively rare greenhouse gas has more effect than increasing the level of a more common species.” This statement is true only if the more common gas is approaching its optical saturation density. This is not true for water vapor. This fact is attested to by the common observation that ground fogs and mists are rapidly dissipated by direct sunlight.
“2) More than 90% of the heat transmitted from the surface to the atmosphere comes in the form of terrestrial infrared radiation. Again, mostly radiation from water molecules. Conduction and enthalpic cycling (condensation, not evaporation) play only very minor roles.”
You’re right, I missed a chunk. But I believe the fraction reradiated is more like 80% (Kiehl and Trenberth, 1997). Most of that is radiated back down again, so convection is (just) the major escape route through the troposphere.
Derek,
Excellent post–although I have a few quibbles. You probably would have addressed them if you were given more space, but here goes:
In your point 6, you state:
1) The Sun warms the Earth’s surface.
2) Conduction and evaporation carry this heat energy to the air at the Earth’s surface.
3) Some conduction, but mostly convection, carries the heat energy higher.
All of these statements are misleading in some respect:
1) Some two-thirds of the radiant energy reaching the surface of the Earth comes from atmospheric molecules—mostly water molecules, the remaining third comes from the Sun. These atmospheric water molecules, of course, got their energy from the Sun in the first place. If you had simply said that, “The Sun warms the Earth”, your statement would be correct.
2) More than 90% of the heat transmitted from the surface to the atmosphere comes in the form of terrestrial infrared radiation. Again, mostly radiation from water molecules. Conduction and enthalpic cycling (condensation, not evaporation) play only very minor roles.
3) Once again, radiation plays the dominant role. Air is simply an extremely poor conductor of heat. An air mass has less thermal energy and a much lower temperature after it has been forced up than it had at its lower elevation. How does this “carry the heat energy higher”?
“… I understood that N2 scatters blue light, and this is what makes the sky look blue. … Buzz”I think that is miss leading. Even if the atmosphere were argon, the sky would still be blue. On the scale of longer wave lengths, say red light wave length, there is nearly a constant number of atoms (or molecules) in a cube with edge of length equal to red wave lengths. But in a cube with half that edge length (1/8 as many molecules inside) the statistical fluctuation as a percentage in number of molecules inside the cube are more significant. The scattering is due to the greater varying index of refraction that small volumes have. Nothing to due with air being mostly N2.
“To address your earlier comment I already added a clause on time delayed feedbacks. If you feel it is necessary to mention instability specifically I’m happy to add that.
Thanks.”
No. I like your insight post. Thanks for the addition. I think what you have is as good as it can reasonable be. Negative feedback such a can of worms that it’s not realistically possible to do much better.
“Negative feedback is more complicated than you imply. It can cause dynamic instability. It depends on the dynamics of the system, which is very complicated in this case.”
To address your earlier comment I already added a clause on time delayed feedbacks. If you feel it is necessary to mention instability specifically I’m happy to add that.
Thanks.
“I included some examples. I made no claim it was exhaustive. There are several more I could have listed (positive and negative). My purpose was to illustrate what is meant by a feedback.
The Insights post concerns aspects of climate science that are well settled and uncontentious yet often misunderstood or misrepresented. I have no plan to get into the more debatable areas. That would be beyond my expertise.
Last I checked, the general view was that whether clouds act mainly as negative or positive feedbacks depended on altitude, but I don’t recall which way it worked or whether there was consensus on which dominates.”Negative feedback is more complicated than you imply. It can cause dynamic instability. It depends on the dynamics of the system, which is very complicated in this case.
“Sure, then perhaps say so? “divided into negative and positive feedbacks, such as …”. Or, “…for example””
Done.
“I included some examples. I made no claim it was exhaustive. …”Sure, then perhaps say so? “divided into negative and positive feedbacks, such as …”. Or, “…for example”
“Regardless of the current uncertainty on the subject, why no inclusion of the possibility of clouds in Negative Feedbacks? The CFMIP modeled feedback is “approximately zero”, but this is not the same as saying there is none.
IPCC AR5 7.2.5.4: “The key physics is in any case not adequately represented in climate models. Thus this particular feedback mechanism is highly uncertain””
I included some examples. I made no claim it was exhaustive. There are several more I could have listed (positive and negative). My purpose was to illustrate what is meant by a feedback.
The Insights post concerns aspects of climate science that are well settled and uncontentious yet often misunderstood or misrepresented. I have no plan to get into the more debatable areas. That would be beyond my expertise.
Last I checked, the general view was that whether clouds act mainly as negative or positive feedbacks depended on altitude, but I don’t recall which way it worked or whether there was consensus on which dominates.
“Minor comment: Please replace that image that depicts a 5250 °C blackbody curve with something else. The Sun’s effective blackbody temperature is not 5250 °C, or 5523 kelvins. It is 5777 or 5778 kelvins, depending on who you read. Siting that image has become a “frequently made error in climate science.” This is a good example of why wikipedia is not a good source.
Otherwise, this is a very nice insight article.”
Thanks D H, I was not aware that was wrong. But I like the image, much clearer than most, so I’ve just added a note. (Is the curve wrong, or just the number? The peak seems to be in about the same place as in other images I found.)
Minor comment: Please replace that image that depicts a 5250 °C blackbody curve with something else. The Sun’s effective blackbody temperature is not 5250 °C, or 5523 kelvins. It is 5777 or 5778 kelvins, depending on who you read. Siting that image has become a “frequently made error in climate science.” This is a good example of why wikipedia is not a good source.
Otherwise, this is a very nice insight article.
The Insight article is excellent. But I can’t help making a small correction to this statement:
“Note: If something is a negative feedback it acts to dampen change; it cannot make the change go in reverse.”
In systems with time delays, accumulated effects, and limiting functions, negative feedback can cause cyclic behavior that includes periodic reverse effects.
The dynamics of the system we are talking about is very complicated and includes all those features.
That being said, if the negative feedback causes a reverse effect at one time, there will probably be a contributing effect later. So it might fool us now and cause a catastrophe later.
“Oh, the horror!! Yet, one must secure government grants or perish.
In most scientific models, a single false prediction invalidates the model.
Climate models seem immune from such scrutiny.”
You are wandering off the topic of what errors are commonly made and what the correct versions are. If you wish to engage in a discussion on the politicisation of science; on the relationship between scepticism, consensus, scientific progress and public policy; or on the use of models of complex systems, let’s do that as a private dialogue.
“some people making climate science errors do so disingenuously.”
Oh, the horror!! Yet, one must secure government grants or perish.
In most scientific models, a single false prediction invalidates the model.
Climate models seem immune from such scrutiny.
“What conclusions can be drawn from such ‘frequent errors”. Who do you think is making such errors and what are the effects? Thanks.”
They are errors I have come across in posts (not just in these forums), and, just occasionally, errors I have made myself.
This particular FME post is a bit different from my others (Friction, Moments, etc.) in that there is reason to suspect that some people making climate science errors do so disingenuously.
What conclusions can be drawn from such ‘frequent errors”. Who do you think is making such errors and what are the effects? Thanks.
“Hi haruspex:
I very much like your Insight article. I have a question though.
The article says:
[INDENT]If we treat the Earth as emitting and absorbing radiation as a “black body”, ignoring the atmosphere, and treating incoming light as spread evenly over the whole Earth’s surface all the time, we can calculate the equilibrium temperature as -18C. At that temperature, black body radiation would balance insolation.
Adding a non-greenhouse atmosphere, e.g. pure nitrogen, doesn’t change this.[/INDENT]
I understood that N2 scatters blue light, and this is what makes the sky look blue. Wouldn’t the scattering of blue photons by N2 then reduce the number of blue photons that hit the earth, since about half the scattered photons return to space? Wouldn’t this [U]reduce[/U] the equilibrium average temperature of the surface compared with no atmosphere?
Regards,
Buzz”
Yes, that’s true too. From what I read, the scattering of green light in air at 1atm. is 10[SUP]-5[/SUP]m[SUP]-1[/SUP], and blue light about double that. From that I calculate that about 4% of green light and 8% of blue light would never make it through. Taking the mean power loss as 6%, that would knock 1.5% off the surface temperature, or 4[SUP]o[/SUP] K.
Edit: I forgot to allow for IR, barely scattered at all. So I would think the power lost would be no more than 4% in total.
Hi haruspex:
I very much like your Insight article. I have a question though.
The article says:
[INDENT]If we treat the Earth as emitting and absorbing radiation as a “black body”, ignoring the atmosphere, and treating incoming light as spread evenly over the whole Earth’s surface all the time, we can calculate the equilibrium temperature as -18C. At that temperature, black body radiation would balance insolation.
Adding a non-greenhouse atmosphere, e.g. pure nitrogen, doesn’t change this.[/INDENT]
I understood that N2 scatters blue light, and this is what makes the sky look blue. Wouldn’t the scattering of blue photons by N2 then reduce the number of blue photons that hit the earth, since about half the scattered photons return to space? Wouldn’t this [U]reduce[/U] the equilibrium average temperature of the surface compared with no atmosphere?
Regards,
Buzz
Bandersnatch, thanks kindly for answering my question!Intuitively, I still find it hard to understand that we, living on the thin crust of the planet, get so little heat from within the endlessly more voluminous and blazingly hot mantle. Particularly because when you descend into a mine, it gets so much hotter so quickly (1 degree F per 70 feet of depth). You'd think that a lot of that energy would find its way to the surface and be dissipated through the oceans and the athmosphere.Do you happen to know how warm (cold) Earth would be if there were no Sun? If 0 Kelvin is the absolute minimum and 287 Kelvin is the current average temperature on Earth's surface, I wonder how much that would be if we only had Earth's core as an energy source.
If the changing polar albedo effect is well understood, why do educated climatologists continually refer to a lowered polar albedo as absorbing more heat as a positive feedback? The polar regions still have a low "average angle of incidence of insolation" (which is what I thought I said). That means little light coming in. On the greybody thing, I did equate mistakenly albedo with thermal emissivity which is only a weak approximation. Albedo refers to the incoming light (visible and short wave infarred) and emissivity the outgoing light (long wave infarred). The two aren't the same, particularly for liquid water which is very complex (varying with surface conditions like wind/wave conditions). But the reciprocity principal means darker surfaces which absorb more also emit more greybody radiation (though that changes with wavelength). Still, a look at outgoing longwave maps makes it clear that ice is a poorer emitter than water. None of your comments change the point that polar regions emit more heat than they absorb, and polar albedo is not a clear positive feedback.
Light does not strike the Earth evenly. Light at the equator (on average) hits at a 90º angle and has full effect. But towards the poles, this drops off. Meanwhile, heat energy radiates more or less uniformly. This causes the poles to be cooler than the equator. It also means melting ice caps decrease the albedo, which actually increases the blackbody radiation, all other things being equal (which they are not, of course). This is complex because of the Earth’s tilt on its axis affects the radiation patterns, which interacts with the seasonal changes in albedo. Add clouds, water vapor, and seasonal temperature variations to get a poorly understood mix of feedback loops. I know it’s poorly understood since few people seem able to remember the sun doesn’t shine in the Arctic in the winter. I’ve been there. It’s dark for months at a time.
Hi, a noob here, so please bear with me as I show my ignorance :) Anyone who has ever spent some time in the depths of a coal mine knows that it can get pretty hot down in Earth’s crust. (Much) Deeper still, the core of our planet has even been calculated to be 10,800 degrees hot (as hot as the Sun’s surface!). Some 90% of the energy required to maintain this temperature comes from the decay of radioactive elements, and if I understand correctly, it is eventually dissipated through (infrared?) radiation at Earth’s surface. I read that the amount of heat caused by this radiation is almost the same as the total heat measured emanating from the planet (http://phys.org/news/2006-03-probing-earth-core.html). I wonder how all this figures in climate models. Climate scientists always seem to talk about light (energy) coming into our atmosphere from the Sun and then being converted into infrared light, which may or may not be trapped as heat in the atmosphere. But how about the (apparently) much more significant amount of infrared radiation coming from within – do today’s models account for that at all? I read the third section above, “Black Body Earth”, which speaks of an -18 degree equilibrium, but I don’t think I understand. Does that or does that not account for this heat from within the planet? I don’t think it does, or else there wouldn’t be places on Earth where it gets even colder in Winter. Or, maybe those colder places can be explained by regional differences in heat conductivity of the planet’s mantle and crust? Thanks in advance for any enlightening comments!
Regardless of the current uncertainty on the subject, why not include the possibility of clouds in Negative Feedbacks? The CFMIP modeled feedback is “approximately zero”, but this is not the same as saying there is none. IPCC AR5 7.2.5.4: “The key physics is in any case not adequately represented in climate models. Thus this particular feedback mechanism is highly uncertain.”