Kinetics Definition and 234 Threads
-

Confusion regarding a chemical kinetics problem
TL;DR Summary: cannot find out error in solution proposed. [![question with rate laws][1]][1] Now the rate law for the reaction (i.e reaction rate) can be written as: $$ R= k[N_2O_5] $$ my main question is, WHAT is this reaction equal to? what I mean here is, whether $$k[N_2O_5]=...- palaphys
- Thread
- Kinetics Reactions
- Replies: 12
- Forum: Biology and Chemistry Homework Help
-
A
Graduate Kirkendall effect: vacancy concentration and pair interaction energy
I am reading the following article on Kirkendall effect leading to the Formation of a hollow binary alloy nanosphere: a kinetic montecarlo study. I am unable to understand or find in references the reasoning to obtain equation (1):- always_learning
- Thread
- Concentration Diffusion Energy Interaction Kinetics Pair
- Replies: 1
- Forum: Atomic and Condensed Matter
-

What's the derivation of the 'Kinetic Equation' in Chemical kinetics?
Hi, I'm following an introduction course to chemistry and I am reviewing the chapter on Chemical kinetics. It's shown that the reaction speed for a certain component of a general chemical equation such as aA +bB <-> cC + dD , might be expressed as v = k[A]m[ B]m. I was wondering where it does... -
M
Biology How Do Stoichiometric Coefficients Affect Reaction Rates?
Hi, I was attempting the following question and don't know how to find the 'reaction rate': "For the reaction the following stoichiometric coefficients have been determined: 2 ##C## react to form 2 ##E## and 1 ##F##. The rate of formation of E is ##1.6\times10^{–4} mol L^{–1} s^{–1}##. What is...- Master1022
- Thread
- Kinetics Reaction
- Replies: 3
- Forum: Biology and Chemistry Homework Help
-
A
Point Kinetics for Linear Insertion of Reactivity
I want to use the point reactor kinetics equations to solve for the power as a function time when a reactivity of gamma*t is added to a reactor that is at equilibrium at time 0. I am also asked to consider the case where the transients are long-lived compared to the lifetime of prompt neutrons...- a1234
- Thread
- Kinetics Linear Point
- Replies: 1
- Forum: Nuclear Engineering
-

Rigid body kinetics -- force on ankle joint
PROBLEM Here from the conservation of angular momentum I found angular velocity just before impact, $$ H_1 = 0 $$ $$ H_2 = I_0\omega + mV_0d $$ $$ H_2 = 66\omega + 76.(1.2).(0.87)$$ $$ H_1 = H_2 $$ $$ \omega = 1.202 rad/s $$ But I couldn't solve it to find joint force. Thanks in advance,- mastermechanic
- Thread
- Body Force Joint Kinetics Rigid body
- Replies: 6
- Forum: Introductory Physics Homework Help
-

Graduate What Is the Meaning of Thermokinetics?
Wikipedia https://en.wikipedia.org/wiki/Thermokinetics says that "thermokinetics" studies the rate of chemical decomposition, whereas a dictionary https://www.yourdictionary.com/thermokinetic gets no further than saying that it combines thermodynamics and kinetics, and a book on it... -
D
(Conceptual- Planar Kinetics) How to maintain the position
Relevant equations : Kindly advise whether this approach is correct, I am not sure whether the friction force should be considered.- daphnelee-mh
- Thread
- Conceptual Kinetics Position
- Replies: 1
- Forum: Introductory Physics Homework Help
-
G
Undergrad Subquantum kinetics- anything to it?
Is there any evidence to support the idea of subquantum kinetics and alternative it provides to the Big Bang? Most publications which touch on it seem to link back to Dr. LaViolette so that seems odd to me. Has this idea been disproven or is there a chance this is a Continental Drift, Alfred...- Garett Kutcher
- Thread
- Kinetics
- Replies: 5
- Forum: Beyond the Standard Models
-

Why Did My Reaction Order Deviate in Chemical Kinetics Lab?
last week i did a chemistry lab. i mixed NaOH 0.03M with equal part of methyl violet and mesured aborbance in a spectrophotometer. then i did the same thing with a 0.05M and methyl violet i did today the graphs and got the k' =-0.1647 for the first solution and k'=-0.4022 for the second... -

Kinetics Problem: Head-On Collision Between Two Cars
Since the mass of both vehicles is the same, it's possible to calculate Ki which happens to be 512,5J and from there, multiply it by 0,25 since 75% of the energy is gone and I end up with 128,125J. Now my problem is that for the velocity, I have: 25,0 + -20,0 = V1 + V2 which is 5,00 = V1 + V2...- Graador
- Thread
- Cars Collision Kinetics
- Replies: 5
- Forum: Introductory Physics Homework Help
-

Reaction kinetics + Gillespie algorithm: Propensity function?
I'm trying to simulate a simple series reaction stochastically using Gillespie's algorithm. I found this file: What is this 'propensity function'? Say for example I have the simple reactions: A --(k1)--> R R--(k2)--> S are these 'propensity functions' the rates (a wild guess)? I mean; α1 =...- maistral
- Thread
- Algorithm Function Kinetics Reaction
- Replies: 1
- Forum: Materials and Chemical Engineering
-

Analytical solution to mono-group kinetics with a ramp of reactivity
Consider the mono-group model and a ramp in reactivity like ##\rho = -\gamma \beta t## The system is $$\frac {dP}{dt} = \frac {\rho - \beta}{\Lambda} P + \lambda C$$ $$ \frac {dC}{dt} = \frac {\beta}{\Lambda} P - \lambda C$$ 1st method: assume that the concentration of precursor doesn't...- dRic2
- Thread
- Analytical Analytical solution Kinetics Ramp
- Replies: 4
- Forum: Nuclear Engineering
-

Time needed for a Force F to accelerate the crate to 2m/s? (Dynamics)
So above is the question. The picture below is my attempt at the solution: However the answer at the back of the book suggest that T=1.929s- bardia sepehrnia
- Thread
- Accelerate Dynamics Force Friction Kinematics Kinetics Mechanics Time
- Replies: 2
- Forum: Engineering and Comp Sci Homework Help
-

Need book recommendation to learn kinematics and kinetics
Hi I am trying to learn kinetics and kinematics: Euler angles Directional cosine matrices Quaternions Inertia tensors The usage of Jacobians in robotics Manipulator kinematics Eulers equations Newton-Euler equations etc My background is in mathematics, so I would like a book which takes a...- Avatrin
- Thread
- Book Book recommendation Kinematics Kinetics Recommendation
- Replies: 1
- Forum: Science and Math Textbooks
-

Can Euler's Formula Illuminate First Order Reaction Kinetics?
Has anyone ever encountered a discussion on the topic of applying Euler's formula exp(i*x) = cos(x) + i * sin(x) to the equation governing first order chemical (and nuclear) reaction kinetics? d[Reactant]/dt = C*[Reactant] -
B
How to calculate kinetics values?
Hello The table below shows some kinetics values for two types of the GOD enzyme, the free and the immobilised. How can I comprehend what these values mean? I want to know how many grams of substrate are 'converted' per sec per mg of enzyme. I assume the substrate concentration plays role so... -

Question about simple kinetics and stoichiometry
Hi, I'm having trouble with this simple reaction (1): $$A + A → I$$ with ##R = kC^2_A## I'm assuming it takes place in a Batch reactor so the mass balance should yield: $$\frac {dC_A} {dt} = -2R = -2kC^2_A$$ But, as I recall (I should be wrong though)... -
D
Chemical Kinetics: velocity and equilibrium
Hi everybody, Given a generic reaction ## Reagents ↔ Products ## the velocity of the reaction is ##R = R_{right} - R_{left} ## To take in account that the velocity of the reaction will slow if approaching the equilibrium we say that $$ R = R_{right}(1 - \frac K K_{eq}) $$ Although it seems...- dRic2
- Thread
- Chemical Chemical kinetics Equilibrium Kinetics Velocity
- Replies: 3
- Forum: Chemistry
-
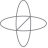
The path of the 3.6-kg particle P is an ellipse
Homework Statement 13.71 The path of the 3.6-kg particle P is an ellipse given by ##R = \frac {R_0} {(1+ecosθ)}##. where R_0 = 0.5m and e=2/3. Assuming that the angular speed of line OP is constant at 20 rad/s, calculate the polar components of the force that acts on the particle when it is at...- Alexanddros81
- Thread
- Ellipse Kinetics Particle Path
- Replies: 3
- Forum: Introductory Physics Homework Help
-

A package of mass m is placed inside a drum
Homework Statement 13.69 A packege of mass m is placed inside a drum that rotates in the vertical plane at the constant angular speed ##\dot {θ} = 1.36 rad/s##. If the package reaches the position θ = 45deg before slipping, determine the static coefficient of friction between the package and...- Alexanddros81
- Thread
- Drum Kinetics Mass Rotation
- Replies: 1
- Forum: Introductory Physics Homework Help
-

The pendulum is released from rest with θ = 30deg
Homework Statement 13.54 The pentulum is released from rest with θ = 30deg. (a) Derive the equation of motion using θ as the independent variable. (b) Determine the speed of the bob as a function of θ. The solutions given in the textbok are a) ##\ddot θ = -4.905sinθ rad/s^2## b) ##6.26\sqrt...- Alexanddros81
- Thread
- Cylindrical coordinates Kinetics Pendulum Rest
- Replies: 5
- Forum: Introductory Physics Homework Help
-

Calculate the angular velocity and angular acceleration (pendulum)
Homework Statement 13.53 The tension in the sting of the simple pendulum is 7.5N when θ=30deg. Calculate the angular velocity and angular acceleration of the string at this instant. Homework EquationsThe Attempt at a Solution Is this correct?- Alexanddros81
- Thread
- Acceleration Angular Angular acceleration Angular velocity Cylindrical coordinates Kinetics Pendulum Velocity
- Replies: 1
- Forum: Introductory Physics Homework Help
-

Banked Curve-Determine smallest static coefficient of friction
Homework Statement 13.49 The flatbed railway car travels at the constant speed of 80 km/h around a curve of radius 55m and the bank angle 15deg. Determine the smallest static coefficient of friction between the crate and the car that would prevent the crate of mass M from sliding. Homework...- Alexanddros81
- Thread
- Banked curve Coefficient Coefficient of friction Friction Kinetics Static
- Replies: 9
- Forum: Introductory Physics Homework Help
-

Determine stopping distance of a train traveling at 20m/s
This is problem 12.81 from Pytels Dynamics 2nd edition 1. Homework Statement A train traveling at 20m/s is brought to an emergency stop. During braking, the acceleration is a=-(7/4)+(t/16) m/s^2, where t is the time in seconds measured from when the brakes were applied. (a) Integrate the...- Alexanddros81
- Thread
- Dynamics kinematics Euler method Kinetics Numerical integration Stopping distance Train
- Replies: 3
- Forum: Introductory Physics Homework Help
-

Calculating Height h from a Projectile Launched at A
Homework Statement A projectile launched at A with an initial velocity of 24 m/s at the angle θ=60deg, impacts the vertical wall at B. Neglecting air resistance, calculate the height h Homework EquationsThe Attempt at a Solution Here is my solution As you can see to find height h I assumed...- Alexanddros81
- Thread
- Dynamics Height Kinematics Kinetics Newton 2nd law Projectile
- Replies: 4
- Forum: Introductory Physics Homework Help
-

Pytels Dynamics 12.39: A rocket is launched vertically
Homework Statement A 2000-kg is launched vertically from the surface of the earth. The engine shuts off after providing a constant propulsive force of 60 kN for the first 20 seconds. Neglect the reduction in the mass of the rocket due to the burning of the fuel and the variation of the...- Alexanddros81
- Thread
- Dynamics Kinematics dynamics Kinetics Newton 2nd law Rocket
- Replies: 3
- Forum: Introductory Physics Homework Help
-

Pytels Dynamics 12.35: A cart carrying a small package
Homework Statement The cart carrying the small 9-N package A is moving up the incline with constant acceleration α. If the package stays at rest relative to the cart in the position θ=45deg, determine the value of α. Neglect friction. Homework EquationsThe Attempt at a Solution [/B] Are my...- Alexanddros81
- Thread
- Cart Dynamics Dynamics kinematics Kinetics Newton 2nd law
- Replies: 3
- Forum: Introductory Physics Homework Help
-
Reactor Design and Kinetics of Reactions
Homework Statement My problem deals with understanding why we substitute in the reaction kinetics equivalent into mass conservation equation instead of dealing with differentials Homework Equations From the conservation of mass law about an envelope: $$\frac{dm}{d t} = \sum m_{in} - \sum...- CivilSigma
- Thread
- Design Kinetics Reactions Reactor Reactor design
- Replies: 9
- Forum: Engineering and Comp Sci Homework Help
-
B
Kinetics question (Chemical engineering)
Hey! I have a question regarding this question that I have to answer. It is about the kinetics of a substance X whose product is 2Y. It is in Swedish and here is the translation: The absorbance of a 0,030M-solution of X was calculated with the help of a Spectrometer. The Spectrometer was...- Beyar
- Thread
- Chemical engineering Chemical kinetics Chemistry Engineering Kinetics Physical chem Physical chemistry
- Replies: 7
- Forum: Biology and Chemistry Homework Help
-
S
Kinetics of System Particle/ Pulley system with masses and i
I can't seem to solve it, considering inertia can someone please solve it, both of the following question in the given picture. I would be very much thankful.- Shehry
- Thread
- Kinetics Particle Pulley Pulley system System
- Replies: 1
- Forum: Mechanical Engineering
-
T
MCQ: Enzyme Kinetics - Find the Zeroth Order Answer
Homework Statement An enzyme catalysed reaction becomes zeroth order when: a.The velocity of enzyme action very low b. The velocity of enzyme action very high c.Enzyme conc very high d. All of these The attempt at a solution I understand An enzyme mediation recation becomes Zeroth order if [...- Tyto alba
- Thread
- Biochemistry Kinetics
- Replies: 1
- Forum: Biology and Chemistry Homework Help
-
L
What equations are needed to solve this wheelchair kinetics problem?
Homework Statement A man is pushing the wheelchair having combined mass of 80kg with center of gravity at CG. The mass of each rear wheel is 1.6 kg and moment of inertia 0.1kgm^2. Friction coefficient for all wheels 0.6 . Find maximum acceleration of the wheelchair without slippage between the...- Lalaland
- Thread
- Kinetics
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-

How to Determine Reaction Kinetics and Conversion Rates for Ammonia Synthesis?
I need to design an ammonia production process but I can't find the kinetic info from the articles online. The catalyst will be iron. Can somebody explain how to find kinetics data and conversion rate? I found some articles but there was no conversion info on them.- Thermo
- Thread
- Ammonia Kinetics Reaction
- Replies: 3
- Forum: Materials and Chemical Engineering
-
C
Iodine Clock Kinetics: Which Mechanism is Supported?
1. Homework Statement An iodine clock kinetics experiment was performed by varying the composition of reactant solutions and measuring resulting rates of reaction (based on the data given, I have determined the rate law to be R = [I-]1[S2O82-]1). Which of these four proposed mechanisms below...- cvc121
- Thread
- Clock Kinetics Mechanism
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-

Fischer-Tropsch Conversion / Kinetics
Hi, We are currently working on a small-scale Fischer-Tropsch process. The modeling software we are using is HYSYS (Aspen). We want to produce X barils of oil / day using syngas (H2:CO ratio 2:1) using cobalt catalysts. The problem we are currently facing is that we do not have the kinetics...- Cyril St-Amand
- Thread
- Hydrocarbon Kinetics Unit conversion
- Replies: 1
- Forum: Materials and Chemical Engineering
-

Undergrad Energy Conversion With Supercritical Fluids
Technically, this isn't a question concerning supercritical fluids. It's more about converting thermal energy into velocity by playing "keep-away" with molecules that want to go supercritical. The idea is simple. In an fully contained system, we can more or less say volume is static...- Benign Paradigm
- Thread
- Energy Energy conversion Fluids Heat Kinetics
- Replies: 5
- Forum: Thermodynamics
-
B
[Kinetics] Using Rate as a Conversion Factor
Homework Statement Certain pollutants encourage the following decomposition of ozone: 2O 3(g) -> 3O 2(g), at a rate of 6.5 x 10-4 M O 3/s. How many molecules of O2 gas are formed in each litre of atmosphere every day by this process? (As this problem provides a rate in units of mol/L/s and...- brbrett
- Thread
- Kinetics Rate
- Replies: 4
- Forum: Biology and Chemistry Homework Help
-
E
How Can I Solve This 2D Kinetics Problem with Time-Dependent Reactions?
Homework Statement Hi, I've been trying to solve this exercise for the last four hours and I'm totally stuck. The problems goes like this: Given the mechanism in the image, located in the vertical plane, the OB cord is cut when t=0. Find the reactions Rx, Ry and N in {Kgf} for the instants...- EzequielJC
- Thread
- 2d Kinetics Rigid body dynamics Stuck
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-

What are the forces acting on a car on a slope at an angle of θ=33.2°?
Homework Statement Please refer to both figures. The result of the example problem was θ=33.2° Homework Equations ΣFn=man The Attempt at a Solution I am stumped. I thought you could solve the forces in the z direction, straight up from the car, and set that to zero since there is no...- Auburn2017
- Thread
- Kinetics Particles
- Replies: 2
- Forum: Introductory Physics Homework Help
-
S
Determining Rate Constant for Hydrolysis of Ethyl Lactate with Acid Catalysis?
Homework Statement Hydrolysis of ethyl lactate Determine the value of the rate constant for the hydrolysis of ethyl lactate from the following data. Mass of ethyl lactate (grams): 0.5956 Concentration of stock HCl solution (M): 0.01023 Standardization titration volumes (of NaOH): Run...- sarahrpol
- Thread
- Chemistry Homework Kinetics
- Replies: 2
- Forum: Biology and Chemistry Homework Help
-
H
Solve Km with Alpha-ChymotrypsinHydrolysis & 4-Nitrophenyl Trimethylacetate
I did a practical and am now trying to do the data analysis of it. the prac was the alpha-chymotrypsin catalyzed hydrolysis of 4-nitrophenyl trimethylacetate. I have values for X, Y and B and the lab manual has asked "Using the approximated, simplified expressions for X (equation 2), Y (equation...- hard0174
- Thread
- Kinetics
- Replies: 2
- Forum: Biology and Medical
-

Kinetics with Pressure: Explanation & Derivation
This post is not about a homework problem but about a derivation When i was flipping through a physical chemistry book I found a formula which looked like How ever I am clueless to what it is Can anyone explain what it is for And possibly Derive it Thank you- sidt36
- Thread
- Kinetics Pressure
- Replies: 4
- Forum: Biology and Chemistry Homework Help
-

Is This the Correct Method to Solve a Kinetics Problem Using Half-Life Formula?
Homework Statement Homework Equations Is the question asking the concentration when the system has reached its half life Can use the half life formula?[/B]The Attempt at a Solution So 1/cT + 1/co = kt 1/(4 *10-2) + 1/x = k(half life time) Is this the right approach Please help me...- sidt36
- Thread
- Chemical kinetics Kinetics Olympiad
- Replies: 3
- Forum: Biology and Chemistry Homework Help
-
M
Reaction Kinetics, batch reactor varying volume
NO TEMPLATE BECAUSE THREAD WAS ORIGINALLY ENTERED INTO WRONG FORUM Hi Guys, I have attached the question that I am struggling with (part b of the question) I have worked out that the conversion of A is X=0.21 Using this conversion I'm trying to work out the time required for the reaction I...- MatStrat
- Thread
- Batch Kinetics Reaction Reactor Volume
- Replies: 1
- Forum: Engineering and Comp Sci Homework Help
-
S
Magnitute and Direction of Electric Field Based on Particle
Homework Statement A positively charged particle initially at rest on the ground accelerates upward to 180 m/s in 2.70 s .The particle has a charge-to-mass ratio of 0.100 C/kg and the electric field in this region is constant and uniform. What are the magnitude and direction of the electric...- Slightly Odd Guy
- Thread
- Charge Direction Electric Electric field Field Kinetics Particle
- Replies: 3
- Forum: Introductory Physics Homework Help
-

What are some examples of kinetic equations and graphs in a kinetics course?
Hey guys, new here :biggrin: Just started a kinetics course and I'm looking forward to discuss the meanings and maybe get some examples of the kinetic equations and graph's I'll encounter (just finished the time distance stuff) pardon my newness to the world of physics, this would be my first...- Marleycake
- Thread
- fun kinetics
- Replies: 3
- Forum: New Member Introductions
-
D
Activation energy and pre exponential factor
Homework Statement Calculate activation energy and pre exponential factor, i need help with the pre exponential factor Homework Equations Temperature °C -20 = 253 k -10= 263 0=273 10=283 20=293 Rate const. s-1 1.15 3.85 11.3 28.7 74.0 The Attempt at a Solution i already plotted the graph on...- Diamond101
- Thread
- Activation Chemistry Energy Exponential Kinetic theory Kinetics
- Replies: 1
- Forum: Biology and Chemistry Homework Help
-

Solids and Liquids in Kinetics and Equilibrium
I just now read in my textbook that solids and liquids are not included in equilibrium expressions, are they included in rate laws? If not, why? Would a greater molar amount of solid or liquid increase the rate of reaction? And why isn't surface area included in Rate Laws? Also, for the... -
T
Help with a mechanics problem from Kleppner's book
Homework Statement An elevator departs the ground at t=0 With uniform velocity. At T1 a boy drops a marbel through the floor of the lift that falls under constant acceleration g=9.8 ms^-2. If it takes marbel T2 time to fall down, what was the height at the T1. Homework Equations h=vT1 h= vT2...- Tahmeed
- Thread
- Book Kinetics Mechanics
- Replies: 1
- Forum: Introductory Physics Homework Help